The American Diabetes Association estimates that over
29 million Americans
— more than nine percent of the population—have been diagnosed with diabetes.
Diabetes
is a metabolic disorder characterized by the inability to let glucose from the bloodstream into cells, where it can be used for energy. The condition is often mentioned in What A Year! stories because it is commonly associated with the development of
obesity,
heart disease,
and some
cancers.
There are two main kinds of diabetes, known as Type 1 and Type 2.
Type 1 diabetes,
previously known as juvenile diabetes, is a chronic disorder usually diagnosed in children and young adults. In patients with Type 1 diabetes, the body does not produce
insulin,
the
hormone
responsible for moving sugars
(glucose)
from the bloodstream into cells. Scientists believe that Type 1 diabetes is an
autoimmune disorder,
which means that the body mistakenly attacks itself, resulting in the destruction of the insulin-producing cells in the
pancreas.
Type 1 diabetes cannot be cured, but it can be successfully managed through insulin therapy and careful monitoring of diet, especially sugar consumption.
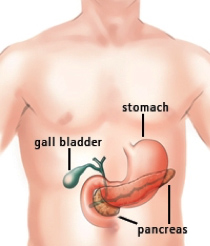
Figure 1. Location of the pancreas in the body.
From: http://columbiasurgery.org/pancreas/pancreas-and-its-functions
Like patients with Type 1 diabetes, patients with
Type 2 diabetes
must also learn to regulate their blood sugar through careful monitoring of diet and exercise routines. Whereas Type 1 diabetes begins in childhood and adolescence, Type 2 diabetes can develop at any time. People with Type 2 diabetes continue to produce insulin, but the body’s cells can no longer use the insulin to effectively regulate blood sugar. This may be due to a problem with the insulin molecule itself or the insulin receptor may no longer recognize the insulin molecule. There is a clear link between sugar consumption, obesity, and diabetes. The development of obesity puts patients at a much higher risk for Type 2 diabetes, for reasons that scientists do not yet understand.
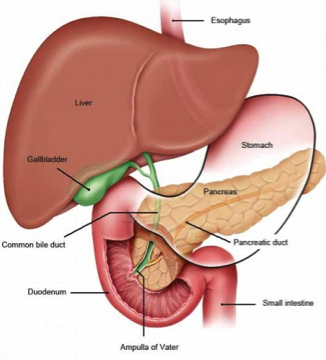
Figure 2. Pancreas and neighboring organs.
From US National Library of Medicine. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0072490/
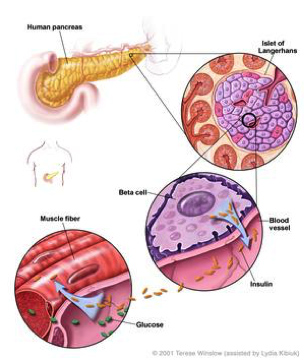
Figure 3. How insulin is produced and works in the body.
From US National Library of Medicine. https://www.ncbi.nlm.nih.gov/pubmedhealth/PMHT0024496/
Although patients with Type 2 diabetes can enjoy long, healthy lives with proper management, there is currently no cure for the disease in the general population. In recent decades, however, scientists have noticed that certain types of weight loss surgery can completely eliminate Type 2 diabetes in patients with severe obesity—even before they have experienced any significant weight loss.
This intriguing finding caught the attention of Dr. Nicholas Stylopoulos, Assistant Professor of Pediatrics at Boston Children’s Hospital specializing in pediatric
endocrinology.
Dr. Stylopoulos focuses on a weight-loss procedure called Roux-en-Y
gastric-bypass surgery.
In gastric bypass, the surgeon first shrinks the size of the stomach by stapling it closed. The small pouch that is left is attached directly to the small intestine, effectively bypassing the digestive function of the stomach. The name Roux-en-Y gastric bypass comes from the Swiss surgeon Cesar Roux, who first described the procedure in 1893. The “Y” in the name, denotes the reconfiguration of the intestines, which after the procedure look like the letter “Y.” The literal meaning of “Roux-en'Y” is “do the intestinal surgery like Roux does it!” After the procedure, the food travels from the esophagus directly into the intestine bypassing the stomach, hence the name “gastric bypass.”
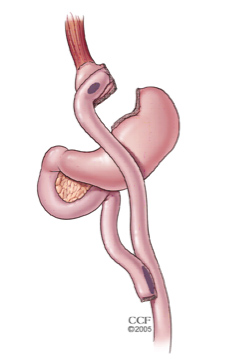
Figure 4. Roux-en-Y gastric bypass.
From American Society of Metabolic and Bariatric Surgery.
https://asmbs.org/patients/bariatric-surgery-procedures#bypass
Gastric bypass surgery is the most successful treatment for obesity and the most successfully performed weight-loss surgery, with many patients losing as much as 90 percent of their excess body weight in the first year alone. It essentially also presents the only known sustainable cure for Type 2 diabetes. It is a common misconception, even among physicians, that gastric bypass works just because the stomach shrinks or nutrients might not get absorbed because of the intestinal reconfiguration. Many recent studies and clinical observations have shown that the mechanisms of gastric bypass surgery are more complex.
Weight-loss surgery is not recommended for the majority of people with obesity or Type 2 diabetes. The risks are simply too high. The procedure must take place under
general anesthesia,
which itself can pose risks to the patient, and the risks of infection and complications are always present. For these reasons, Dr. Stylopoulos set out to understand the mechanisms driving the successful elimination of Type 2 diabetes after surgery, in the hope that he could realize the same results in patients without a surgical procedure.
Dr. Stylopoulos and his team of colleagues began with animal models of obese, diabetic mice and rats. The researchers performed the same gastric bypass surgery on the rodents that has been done in humans, and they achieved the same results—shortly after completion of the procedure, the diabetes in the rodents also cleared. To understand how this was happening, the researchers examined glucose metabolism in specific organs using an imaging technique known as positron emission tomography-computer tomography
(PET-CT).
Prior to the PET-CT scan, the rats and mice were injected with a fluorescent tracer for glucose. The images obtained provided a picture of where glucose was utilized in the body and the rate of utilization. The researchers performed the scan twice: once just prior to surgery and once immediately following surgery.

Figure 5. PET-scan with (right) and without (left) the gastric bypass surgery. On PET scan, glucose uptake is color-coded. The higher the intensity of the color, the higher the uptake. Some organs (e.g., brain) always have an intense signal. However, the abdomen and the intestine are usually areas with low signal (e.g., on the left without the surgery). After the surgery (right), there is significantly more signal and thus more glucose uptake in the intestine.
Using these imaging techniques, the researchers honed in on the small intestine, specifically the middle portion that, after surgery, becomes the first area to receive food. Not only did the small intestine exhibit an increase in glucose metabolism after surgery, the researchers observed that the organ had actually increased in size. “The small intestine becomes a different organ after surgery,” explains Dr. Stylopoulos. “It becomes bigger and works harder because the environment has changed.” The environment Dr. Stylopoulos refers to includes the mechanical forces in and around the intestines, the acidity inside the intestines, as well as the types and numbers of microbes.
The researchers continued to study cellular function in the intestine to determine the mechanisms leading to the increased glucose metabolism they observed. Chemical assays revealed increases in all the products and by-products of
glycolysis,
the process by which glucose is converted into energy useful for the body’s cells. In addition, gene expression analysis showed increased expression of
enzymes
involved in glycolysis. Specifically, the researchers identified an increase in the expression and presence of the protein GLUT-1, which is responsible for transporting glucose from the blood into cells.
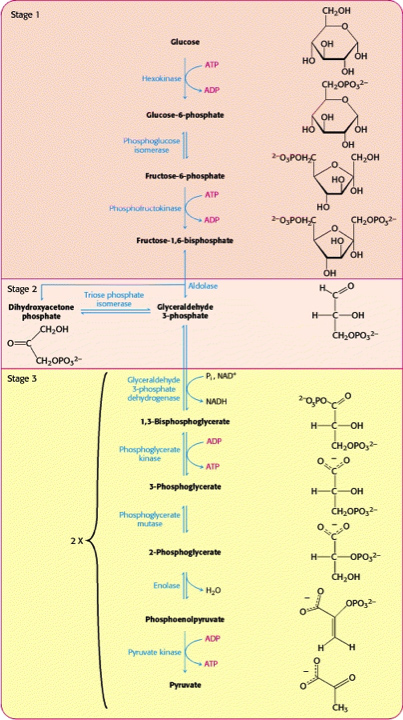
Figure 6. Glycolysis is an energy conversion pathway.
From Biochemistry – National Library of Medicine. https://www.ncbi.nlm.nih.gov/books/NBK22593/
The increased level of GLUT-1 in the intestines is one reason why the post-surgery rats and mice utilized increased amounts of glucose. This had a direct effect on blood glucose levels and the presence of diabetes.
What was it about the post-surgery environment that caused these changes? One hypothesis is the increased biomass of microbes in the intestines. “Microbes may trigger processes in the intestine that depend upon glucose,“ explains Dr. Stylopoulos, “or it may be the different pH or the change in the mechanical forces, the sheer stress of the impact of the undigested nutrients on the intestinal wall. What is definitely clear is that the bioenergetic needs of the intestines are fundamentally altered by this surgery.”
In order to better understand their findings, Dr. Eirini Nestoridi, a research scientist in Stylopoulos’ lab spearheaded an effort to search the scientific literature for other examples of intestinal growth. One in particular stood out, conducted by a team of biologists led by Dr. Stephen Secor at the University of Alabama entitled “A vertebrate model of extreme physiological regulation.” Dr. Secor’s research focused on the digestive systems of a variety of animals, and in one study his team reported on an extreme increase in intestinal size in the reptilian species Python bivittatus, more commonly known as the
Burmese Python.

Figure 7. Burmese pythons in the lab.
Burmese pythons can survive for months without consuming a single meal. As a result of disuse, the entire digestive system goes into a hibernation-like state and the intestines become very small. Upon feeding, however, the intestines expand to six times their previous size, a process that begins within six hours after feeding and, within three days, reaches its largest size. Two weeks later, the intestine has shrunk back to its previous, small size. In 2008, Dr. Secor and his team conducted PET scans on fasting and fed pythons and reported significant increases in glucose utilization in the intestines.
Extremely interested in this natural model of intestinal growth, Dr. Nestoridi and Dr. Stylopoulos began to think about how the lab might obtain some pythons to study in Boston. They were helped in this endeavor by Dr. Arthur Nedder, a veterinarian at Children’s Hospital who loves snakes and had lots of experience with their care. Dr. Nedder taught many members of the team to handle the pythons and feel comfortable around them. “Dr. Nedder was absolutely critical to this project,” comments Dr. Stylopoulos. Boston Children’s Hospital is now one of only three institutions in the United States to study pythons for medicinal purposes.
Dr. Stylopoulos and his team analyzed gene expression before and after meals in ten pythons. They observed an increase in glycolysis in the intestines, just as they had observed in mice. This indicates a common metabolic pathway between the two species. There is anecdotal evidence that similar processes are at work in humans, but to understand the mechanisms behind these observations human studies are necessary.
To obtain human data, Dr. Stylopoulos and his team are working with gastric bypass surgeons and patients. Patients who agree to participate in the study allow the researchers to take a sample of intestinal tissue before or at the time of surgery, one month after surgery, and six months after surgery. The results of this long-term study are still preliminary, but so far the researchers have shown that the common pathways identified in pythons and rodents are also involved in the intestinal increase observed in post-surgical human patients.
There is still much research ahead, but the scientists are hopeful that their findings will lead to non-invasive treatments for Type 2 diabetes. “We are looking not just at the management of Type 2 diabetes but the potential elimination of this disease from the body,” concludes Dr. Stylopoulos. “This is an exciting time for diabetes research.”
Dr. Nicholas Stylopoulos is Assistant Professor of Pediatrics at Boston Children’s Hospital and Harvard Medical School. His research focuses on the mechanisms behind effective weight-loss and the reduction of co-morbidities as a result of weight-loss surgery in conjunction with the Center for Basic and Translational Obesity Research. When not in the laboratory, Dr. Stylopoulos enjoys biking, swimming and spending time with his family.
For More Information:
- Saeidi, N. et al. 2013. “Reprogramming of intestinal glucose metabolism and glycemic control in rats after gastric bypass.” Science, 34(6144): 406-10.
To Learn More:
- Stylopoulos laboratory.
http://www.childrenshospital.org/research-and-innovation/research/labs/stylopoulos-laboratory
- American Diabetes Association.
http://www.diabetes.org/
- Centers for Disease Control and Prevention.
http://www.cdc.gov/diabetes/
- Gastric Bypass Surgery.
https://medlineplus.gov/ency/article/007199.htm
Related What A Year Stories
- Sugar High.
http://www.whatayear.org/04_09.html
- Honey I Shrank the Sensor.
http://whatayear.org/09_09.html
- Get Up and Move.
http://whatayear.org/05_07.html
- Early Detection Saves Lives.
http://whatayear.org/04_16.php
- Microscopic Magnets.
http://whatayear.org/05_15.php
- Let’s Get Specific.
http://whatayear.org/01_12.php
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com

