|
Did you know that the first
magnets
were discovered over four thousand years ago? It was an accidental discovery—metallic objects would stick to mysterious black rocks called
lodestones.
We now know that lodestones contain
magnetite,
a naturally magnetic compound made from iron oxide, Fe3O4. Since their discovery in ancient times, magnets have been used for all kinds of purposes ranging from compasses to computers to those little magnets you stick on the fridge. This month we will introduce some of the tiniest magnets imaginable, and explain how researchers are using these microscopic magnets to fight cancer.
In clinical medicine, magnets and
magnetic fields
are used in a variety of imaging techniques, most notably magnetic resonance imaging or
MRI.
MRIs are a common diagnostic tool powered by strong magnets and found at most clinics and hospitals. In a typical MRI scan, the patient is placed inside a human-sized magnetic tube as radio-frequency waves are directed at the body. In the presence of a strong magnetic field, the radio waves give off signals that can be used to create images of internal structures in the body and differentiate between tissue types.
When using such a powerful magnet, certain precautions must be taken. Most importantly, all metal objects must be removed from the body. Patients with implanted devices that contain
ferromagnetic material
(iron or iron alloys), such as a
pacemaker,
cannot undergo an MRI scan. This is because the magnetic field of the MRI will cause the metal device to either move or, in some cases, to reach extremely high temperatures. Metal objects in contact with the human body that undergo magnetic field exposure could create very serious damage by becoming extremely hot or by moving through critical tissues.
Although magnetic heating at the macro level can become deadly, at the micro level it could prove potentially life-saving.
Magnetic heating on the micro level is the focus of new research by an interdisciplinary team of scientists led by Dr. P. Jack Hoopes of Dartmouth College in Hanover, NH. It has been known for several decades that tissues with slightly elevated temperatures respond better to
radiation
and
chemotherapy,
the mainstays of modern cancer treatments. Dr. Hoopes’ research uses
nanotechnology
to harness magnetic heating on a microscopic scale, producing novel therapies that could be used together with existing cancer treatments.
Think small
really small
Let’s begin by thinking very small. The head of a pin is about one millimeter in diameter, or one thousandth of one meter. (For reference, the height of most humans ranges between one and two meters—about three to six feet.) Things measured in millimeters are still visible to the naked eye. Now imagine one millionth of one millimeter, or a millionth of a thousandth of a meter. This is how small one nanometer is. A single human hair is between 60,000 and 120,000 nanometers; a red blood cell is 70,000-80,000 nanometers. A strand of human DNA is about two nanometers—that is two millionths the size of the head of a pin!
Over the years, What A Year! has highlighted several researchers working with technology on the nanometer scale, known as nanotechnology. For example, in November 2012 we learned about Dr. Evgeny Katz and his work with nanotubes to create bio-powered batteries. In September 2009 we featured Dr. Heather Clark and her research into nanosensors to create a diabetes tattoo.
Like those researchers, Dr. Hoopes works with particles on the nanoscale. Cancer cells themselves are small. “Over one million cancer cells can fit on the head of a pin,” explains Dr. Hoopes. “We want to get as many nanoparticles as possible in every cancer cell. However, depending on the type and size of iron oxide nanoparticles we use and how we deliver and target the nanoparticles we believe we can be effective with as few as 15,000 to 20,000 nanoparticles per cell.” These are not just any particles. The nanoparticles Dr. Hoopes refers to are ferromagnetic, and therefore will increase in temperature when exposed to a magnetic field.
First, Dr. Hoopes and his team had to construct the magnetic nanoparticles. To do this, they covered particles of iron oxide—the very same molecules found in lodestones of ancient times—with a carbohydrate coating (dextran). They hypothesized the iron oxide nanoparticles would be safe for combining with radiation and chemotherapy treatments since similar nanoparticles have been used in medicine to treat iron-deficiency anemia and as a contrast agent for MRI imaging.
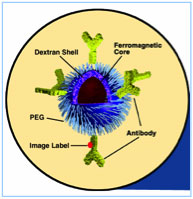
This figure represents the iron oxide nanoparticle we use. It has a ferromagnetic iron oxide core and dextran and polyethylene glycol (PEG) coatings. The figure also shows potential antibody tagging and a fluorescent imaging label.
If enough of these particles enter the cancer cells, Dr. Hoopes and his team have shown that the resulting heat will selectively kill cancer cells. If the intracellular nanoparticle heating is combined with radiation or chemotherapy the level of heat can be reduced.
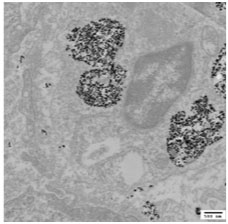
This electron micrograph demonstrates many iron oxide nanoparticles aggregated within an endosome inside a cancer cell.
Around the time that Dr. Hoopes was developing the magnetic nanoparticles, he was approached by a veterinarian concerning a patient with an incurable tumor. It was a locally invasive, generally incurable cancerous tumor, that originated in gum tissue around the teeth. Presented with an opportunity to test the technology on patients in need, Dr. Hoopes and his team developed an experimental procedure, which was approved, and treated the dog’s cancer with magnetic nanoparticles.
Given the success of the first treatment in a dog, Dr. Hoopes and this team continued to treat dogs while simultaneously working to improve the technology.
The nanoparticles were injected directly into the tumor, and after several hours the dog was exposed to a magnetic field similar to an MRI scan. After two treatments, the tumor was effectively killed. It has been over three years since the treatment, and the patient is still cancer free and healthy.
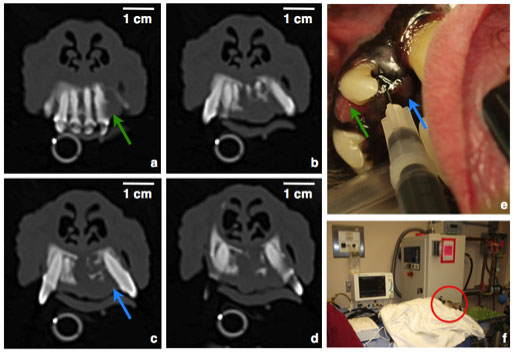
This photo is a CT scan of Jenny’s nasal and hard palate region showing the tumor invading from the peridental region through the hard palate and bone of the nasal floor. The upper right photo (in the series below) demonstrates injection of the nanoparticles into the tumor. The lower right photo shows “Jenny” being treated in our magnetic field delivery facility.

This dog “Jenny” Orndorff was the first dog we treated with iron oxide nanoparticle cancer therapy. She had a tumor in her oral incisor region that is typically incurable with radiation or surgery (Ananthomatous Amelioblastoma). She is now cancer free 40 months after treatment. This a photo of her taken 24 months after treatment.
Given the success of the first treatment in a dog, Dr. Hoopes and this team continued to treat dogs while simultaneously working to improve the technology. Currently the canine treatments are confined to animals with oral tumors because they are easily visible and the nanoparticles can be injected directly into the affected areas. Two types of tumors are eligible for inclusion in the experimental treatment:
Acanthomatous Ameloblastoma
and
oral melanoma.
The ameloblastoma tumor is a locally invasive tumor that expands into surrounding soft tissue and bone tissue, such as the jaw and
maxilla.
This type of tumor does not metastasize to other tissues around the body. Surgery and radiation eliminate the tumor for relatively short periods, but do not cure the cancer. Oral malignant melanoma, on the other hand, is one of the most aggressive and lethal canine cancers. Almost all dogs die from metatstaic cancer within one year of diagnosis, regardless of the type or extent of treatment, surgery, radiation and/or chemotherapy.
In order to be considered for treatment, the patient is almost always referred by the veterinarian who provides its regular care. The Dartmouth research team, which includes two veterinarians, six PhD scientists and several physicians who specialize in cancer therapy, talks with the referring veterinarian and meets with the dog’s owner to explain the treatment. The dog undergoes computerized tomography or CT and MRI imaging scans to locate and determine the extent of the tumor before treatment begins.
Canine cancer treatment
To treat the dog, nanoparticles are either injected directly into the tumor or administered into the blood vessel leading to the tumor in the form of a fluid that looks like black ink. The nanoparticles filter into the tumor over several hours, and the tumor is then exposed to a magnetic field. This is the same concept as an MRI, but the magnetic field is stronger and more focused. The dog is positioned so the affected area is closest to the magnet. While the treatment is going on, the researchers measure tumor temperature and pressure, and other tissue or blood markers that indicate direct tumor damage or an immune reaction, which could help control tumor growth. The treatment is completed within one or two hours, and the dog goes home with its owners at the end of the procedure. The researchers follow up with the patient one week later.

Nanoparticle research team preparing to treat a canine tumor patient in the alternating magnetic field (AMF) suite. The anesthetized patient is placed on the AMF coil prior to injection of nanoparticles into the tumor. Personnel (left to right), Aditi Misra (Dartmouth graduate student), James Petryk (research engineer) Dr. Hoopes and post-doctoral research fellow Dr. Alicia Petryk.
So far the team has treated fourteen dogs. All patients have responded to the treatment and exceeded the cancer remission time afforded by standard treatments. In fact, all four patients with the Acanthomatous Ameloblastoma tumors are still alive and cancer free. The melanoma tumors have proven more difficult, due to the fact that they had aleady started to spread before the patient arrived at Dartmouth for the experimental therapy. Although seven of ten melanoma patients have died from cancer related complications, all of the treated dogs have lived and been cancer free longer than oral melanoma patients receiving conventional surgery, radiation and/or chemotherapy treatments.
Though it has proven very effective so far, treatment with magnetic nanoparticles is currently limited to just a few externally observable tumor types. This is because in the early phases of this new treatment nanoparticle delivery is more effective when the tumor is visible and readily accessible. Two additional nanoparticle delivery methods being tested by the Dartmouth researchers are 1) delivery of cancer-targeted nanoparticles into the blood stream or 2) by running a catheter through the blood vascular system to the tumor region, and delivering the nanoparticles at that point.
Cancer antibodies help targeting
For the treatment to be most effective, the researchers have learned that both the nanoparticle and magnetic field need to be localized to the tumor region. In order to make the nanoparticles as selective as possible towards cancer tissues, the researchers are developing antibody-targeted nanoparticles specific to cancer cells.
Antibodies
are proteins produced by the
immune system
that seek out and bind to particular cells. By attaching specific cancer antibodies to the magnetic nanoparticles, the researchers hypothesized that the antibodies would direct the nanoparticles to the targeted cancer cells.
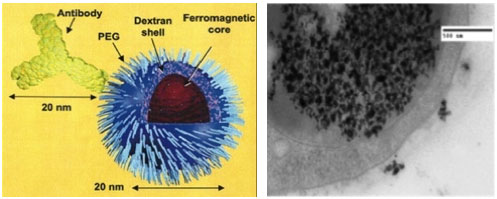
The left side of this figure demonstrates a diagram of our nanoparticle, with various coatings. The right side of this figure is an antibody and electron micrograph showing nanoparticles in an endosome inside the cell and outside the cell, on the cell membrane, ready to be endocytosed.
In order to test the antibody-directed magnetic nanoparticles, Dr. Hoopes and his team used a mouse model of breast cancer. The researchers chose this animal model because they are able to grow various types of breast cancer in mice. These different types of cancers have different antibody binding levels, which enables the researchers to test the effectiveness of antibody nanoparticle targeting to the different cancer cells.
In initial experiments, Dr. Hoopes and his team injected the nanoparticles into the bloodstream and then, to determine safety, monitored their concentration in different tissue types. In the next set of experiments, the researchers attempted to treat the cancer by exposing the mice to a magnetic field, which is similar to the field used in human MRI scanners.

Mouse positioned in the coil that delivers the magnetic field. The treatment is painless, and the mouse is under anesthesia during the treatment.
The results of these experiments have been important for a number of reasons. The researchers found that tumors grown in mice can be effectively treated with antibody-directed nanoparticles and a non-invasive magnetic field. The experiments have also shown that antibody tagged nanoparticles are taken up or retained in tumors 2.5 times more than non-antibody tagged nanoparticles following
systemic administration.
“This is a step in the right direction,” Dr. Hoopes comments. “But I think we can do better.” Experiments to improve the specificity of antibody-directed nanoparticles are ongoing.
Other areas of ongoing research are the use of the magnetic nanoparticles combined with radiation and/or chemotherapy to kill cancer cells. The researchers believe their data demonstrate that magnetic nanoparticles, in combination with other therapies, could significantly increase the effectiveness of current human cancer treatments.
Dr. Hoopes and his team have tested the combination of magnetic nanoparticle and the chemotherapy
cisplatin,
one of the first chemotherapy drugs used to treat breast cancer. While cisplatin has been shown to be effective against breast tumors, like all chemotherapy drugs, its treatment potential is limited by toxic side effects. Dr. Hoopes hypothesized that selective heating of individual cancer cells could improve the cancer selectivity of cisplatin. The researchers used mouse models of breast cancer to test their hypothesis. The magnetic nanoparticle heating plus the cisplatin treatment was approximately twice as effective as either treatment alone.
Dr. Hoopes has observed similar synergism in his dog patients. In addition to the treatment with magnetic nanoparticles, some dogs also received radiation treatment. The researchers found that dogs given radiation treatment after the injection of nanoparticles (but no magnetic field) responded better than those treated with radiation alone. Further study revealed that the addition of the magnetic field produced an even better response. Additional experiments have shown that radiation given before the magnetic nanoparticles improves the uptake of systemically administered nanoparticles by the tumor cells. Radiation changes the microenvironment of the tumor cells, leading to increased nanoparticle uptake,” explains Dr. Hoopes.
The future for animals and people
Dr. Hoopes is optimistic about the therapeutic benefits of his research. Already the magnetic nanoparticles have significantly extended the lives of animal patients. He plans to continue treating animal patients while simultaneously conducting the experiments that will enable his team to improve treatments. “Ultimately, our goal is to treat a variety of tumor types in animal patients—we, of course, are passionate about helping the animals, but also know that the information we learn will directly prepare us to perform these treatments in human cancer patients.”
Dr. P. Jack Hoopes is Professor of Surgery, Radiation Oncology at the Geisel School of Medicine and Adjunct Professor of Engineering at the Thayer School of Engineerring, Dartmouth College. He also serves as Director for the Surgery and Radiation Research Laboratories,the Center for Comparative Medicine and Research and the Small Animal Imaging, Radiation and Microscopy Resource for Dartmouth-Hitchcock Norris Cotton Cancer Center. Dr. Hoopes is a veterinarian with specialty training in pathology.
After graduating from veterinary school, Dr. Hoopes completed a residency in zoo medicine at the National Zoo in Washington D.C. and a residency in veterinary pathology at Colorado State University. While in Colorado, Dr. Hoopes performed pathology studies on many cancer patients and decided to pursue a PhD degree in cancer and radiation research. He completed a post-doctoral fellowship in radiation oncology at Duke University and North Carolina State University before taking a position at Dartmouth College. “It has not been a typical career path,” Dr. Hoopes reflects. When not in the laboratory, Dr. Hoopes enjoys traveling, hiking , running and other types of endurance exercise, working outside, and spending time with his family.
For More Information:
- Pearce, J. et al. 2014. “Magnetic Heating of Nanoparticles: The Importance of Particle Clustering to Achieve Therapeutic Temperatures.” Journal of Nanotechnology in Engineering and Medicine, 4: 110071-14.
- Petryk, A.A. et al. 2013. “Magnetic nanoparticle by hyperthermia enhancement of cisplatinum chemotherapy cáncer treatment.” International Journal of Hyperthermia, 29(8): 845-51.
- Hoopes, P.S. et al. 2013. “Imagine and modification of the tumor vascular barrier for improvement in magnetic nanoparticle uptake and hyperthermia treatment efficacy.” Proceedings of the International Society for Optics and Photonics, 8584.
- Giustini, A.J., A.A. Petryk, and P.J. Hoopes. 2012. “Ionizing radiation increases systemic nanoparticle tumor accumulation.” Nanomedicine, 8(6): 818-821.
To Learn More:
Cancer Treatments
- American Cancer Society. http://www.cancer.org/treatment/treatmentsandsideeffects/treatmenttypes/treatment-types-landing
- National Cancer Institute. http://www.cancer.gov/cancertopics/treatment
Canine Cancer
- National Canine Cancer Foundation. http://www.wearethecure.org/
- American Society for the Prevention of Cruelty to Animals. http://www.aspca.org/pet-care/dog-care/cancer
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |

