| |
You may have heard of a disease called cancer. In 2005, over 11 million people were living with the disease in the United States alone, and it is estimated that 40 percent of people alive in the United States today will be diagnosed with some form of cancer in their lifetime.
Cancer is not one disease but many. The term cancer refers to the condition in which normal cells become unregulated and reproduce out of control. These cells form large clusters called tumors. Some tumors, known as benign tumors, do not interfere with other healthy cells and can simply be removed. Some of the time, though, tumor cells will invade healthy tissue and begin to take over healthy organs. These tumors are called malignant tumors, also known as cancer. Sometimes the malignant tumor cells spread to other parts of the body. When a malignant tumor spreads and grows in another tissue it is known as metastasis.
A Theory of Cancer
There have been many theories about cancer over the years. It is difficult to generalize about cancer, though, because each type of cancer is different. In the early 1970s, the late Dr. Judah Folkman, then a professor at Harvard
University, came up with an idea that would revolutionize the way we think of cancer. Tumors need new blood vessels in order to grow because cells in all tissues, including tumors, require nutrients from the blood supply. In order to thrive, tumors must create their own blood vessels. This process is angiogenesis. If Dr. Folkmans theory were correct, it would mean that by preventing tumor angiogenesis preventing tumors from growing their own blood vessels you could cut off the nutrition source for the tumor and kill it.
A Fortunate Problem
To test his theory, Dr. Folkman isolated cells (called endothelial
cells) from the outer walls of small capillary
blood vessels and cultured them in plastic dishes that he could use for
experiments. One of Dr. Folkmans associates, a young scientist named Dr.
Donald Ingber carrying
out his postdoctoral training in the lab, made an interesting observation one
day while culturing these cells . He went to check on his endothelial cell cultures
and found that the cells were contaminated with a fungus that caused the cells
to round and die. This is a very bad thing: contaminated cells cannot be used
for experiments, and infections can spread in culture dishes just as in humans.
The normal procedure is to just throw out the cells and start over. Instead of
throwing out the contaminated cells, though, Dr. Ingber isolated the fungus and
left it to grow. His basic research had suggested that cell rounding can inhibit
capillary cell growth. When Dr. Ingber and Dr. Folkman later tested molecules
produced by this fungus, they found that they killed the endothelial cells.
The results of Dr. Ingbers experiment with the fungus had important results. Dr. Ingber hypothesized that if this fungus managed to kill the capillary endothelial cells in culture, it might be able to kill them in tumors in animals. If the fungus could kill the capillary cells, it would cut off the flow of nutrients to the tumors and, according to Dr. Folkmans theory, kill the tumor. To test their theory, the two men injected the fungus into mice with tumors. If the theory was correct, the tumors in the mice given the fungus should shrink as their capillary cells (and blood supply) were killed. This is exactly what Dr. Folkman and Dr. Ingber found.
After testing the fungus on tumors in mice, the two men worked with pharmaceutical companies to make the fungus into a drug that could be tested. The drug, known as TNP-470, went into clinical trials in 1992 and it hindered tumor growth in a significant portion of patients. The clinical trials were stopped, however, because, along with stunted tumor growth, the drug also caused neurotoxicity. Somehow this fungus was getting into the brain and creating memory loss and other neurological problems.
Problem Leads to Opportunity
A solution to this problem was recently developed by Dr. Ofra Benny, an expert in drug-delivery systems, in Dr. Folkmans lab. Dr. Benny wanted to create a drug based on TNP-470, with several modifications. The drug would have to be altered to eliminate neurotoxicity, and Dr. Benny also wanted the drug to be administered orally (such as a pill that would be taken by mouth). The original drug had to be administered with a one- to four-hour injection at the hospital several times per week. It was not comfortable for the patients, nor was it practical as a long-term solution to suppress angiogenesis in tumors. An oral drug, on the other hand, could be taken in the patients own home and would be much less invasive.
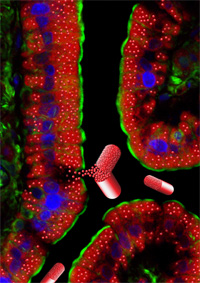 Illustration of an oral drug in the form of nanoparticles being absorbed in the intestine.
The first thing Dr. Benny did was to experiment with administering the TNP-470 drug orally in its original form. The drug was not effective, which is a common problem among cancer drugs. When a drug is given as an injection, it goes directly to its intended destination in the body via the bloodstream. An orally administered drug, however, must first pass through the digestive system before it can be absorbed. The high acidity in the stomach deactivates many drugs, rendering them ineffective. The drug would somehow have to be protected from digestive juices in the stomach but still be able to be absorbed in the small intestine. Illustration of an oral drug in the form of nanoparticles being absorbed in the intestine.
The first thing Dr. Benny did was to experiment with administering the TNP-470 drug orally in its original form. The drug was not effective, which is a common problem among cancer drugs. When a drug is given as an injection, it goes directly to its intended destination in the body via the bloodstream. An orally administered drug, however, must first pass through the digestive system before it can be absorbed. The high acidity in the stomach deactivates many drugs, rendering them ineffective. The drug would somehow have to be protected from digestive juices in the stomach but still be able to be absorbed in the small intestine.
Polymers to the Rescue
Dr. Benny turned to polymers as a possible solution. A polymer is a large molecule made up of many repeating smaller molecules. Her idea was to make a very tiny particle, called a nanoparticle, out of the polymers. Nanoparticles are particles that are measured on the scale of tens of nanometers. To give you an idea of how small a nanometer is, think of it this way: the relationship between a nanometer and a meter is the same as the diameter of a blueberry to the diameter of the earth.
Dr. Benny used polymers that had already been approved by the U.S. Food & Drug Administration. This meant that the polymers were already being used in other drugs and would be guaranteed to be non-toxic. Dr. Benny chose two polymers called polyethylene glycol (PEG) and polylactic acid (PLA). PLA is a hydrophobic, or water-fearing, molecule, which means that it does not dissolve in water. PEG, on the other hand, is a hydrophilic, or water-loving molecule, which means that it does dissolve in water. When these two polymers are put in water along with the TNP-470 drug, said Dr. Benny, a remarkable thing happens. The polymers form a capsule around the drug, creating a natural nanoparticle. The TNP-470 drug is on the inside of this molecular cocoon surrounded by one layer of the hydrophobic PLA and a second layer of hydrophilic PEG that interacts with the water. Dr. Benny called her new creation Lodamin.
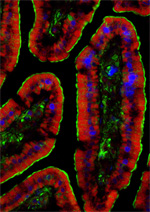 Mouse small intestine: Mouse small intestine:
red fluorescent labeled nanoparticles;
green intestine tissue;
blue cell nucleiWith the creation of Lodamin, the next step was to begin
testing. First Dr. Benny tested Lodamin in vitro, in capillary cell cultures,
in order to see if TNP-470 in its new form was still effective. These capillary
endothelial cell cultures were the same cells that Dr. Folkman had been culturing.
Dr. Benny labeled the nanoparticles with florescent dye so they could be seen
easily. Then she watched as Lodamin was quickly absorbed into the cells. The
rapid absorption rate was a promising sign that the drug would be successful.
Dr. Benny then looked at the cells themselves and saw that Lodamin had inhibited
cell growth. As a control, Dr. Benny tested the endothelial cells with a polymer
that lacked the TNP-470 inside to make sure that the polymer itself was not
affecting the cells responses. The control cultures did not show the inhibited growth that the treatment group had. This indicated that it was the TNP-470 drug, and not the polymer, that inhibited capillary cell growth.
Dr. Benny continued her experiments with Lodamin using mice with tumors. She used two tumor models, lung cancer and melanoma, which are both very aggressive types of cancer. The mice were injected with the cancer cells and then Dr. Benny waited for the tumors to grow. When the tumors reached a certain size, she began the treatment, administering Lodamin orally to the mice. We followed the growth of the tumors, said Dr. Benny, and what we saw was a very dramatic inhibition of tumor growth. She analyzed the tumor tissues and found that one major reason the tumors shrank was the inability of the blood vessels to grow and reach the tumor. This meant that the drug was blocking the blood supply to the tumor and killing it, just as Dr. Folkman had theorized years earlier.
  Illustrations of polymeric micelle structure a nanoparticle with the hydrophobic drug (in gray) in the center, surrounded by the protecting layer of PEG-PLA polymer (in green)As part of the experiments, Dr. Benny looked at how TNP-470 escaped from the Lodamin polymer to be released into the body. Most of the Lodamin nanoparticles congregate at the tumor site. Once there is a high concentration of nanoparticles, the TNP-470 drug will begin to diffuse into the tumor bed. The release of TNP-470 occurs in a slow manner because it must diffuse through two layers of polymers. Dr. Benny observed that it took around 25 days for the full amount of the drug to be released out of the nanoparticle. This is actually a very good thing, said Dr. Benny, because if the release time is so slow, it means the drug will not have to be administered as often. Illustrations of polymeric micelle structure a nanoparticle with the hydrophobic drug (in gray) in the center, surrounded by the protecting layer of PEG-PLA polymer (in green)As part of the experiments, Dr. Benny looked at how TNP-470 escaped from the Lodamin polymer to be released into the body. Most of the Lodamin nanoparticles congregate at the tumor site. Once there is a high concentration of nanoparticles, the TNP-470 drug will begin to diffuse into the tumor bed. The release of TNP-470 occurs in a slow manner because it must diffuse through two layers of polymers. Dr. Benny observed that it took around 25 days for the full amount of the drug to be released out of the nanoparticle. This is actually a very good thing, said Dr. Benny, because if the release time is so slow, it means the drug will not have to be administered as often.
Smart Nanoparticles?
There is still the question of how the nanoparticles know to congregate at the tumor site. Although the nanoparticles do not have any special molecule that recognizes tumor cells, the Lodamin nanoparticles passively gather at tumor sites because of an effect known as the Enhanced Permeability and Retention effect, or EPR effect for short. Tumor blood vessels are very abnormal, Dr. Benny explained. They are very leaky and allow more particles in and out, so naturally the nanoparticles accumulate there.
Next, Dr. Benny continued her experiments in mice with liver cancer caused by metastases. Since the drug was entering the digestive system, large quantities would end up in the liver. Dr. Benny wanted to see if Lodamin could work against liver cancer because very few drugs on the market today can effectively combat the disease. Dr. Benny injected tumor cells into the spleen, where they traveled through the bloodstream to the liver. After the cells reached the liver, they quickly grew into cancer nodules. Once the cancer had reached a certain size, Dr. Benny again began the Lodamin regimen. After three weeks of treatment, Dr. Benny conducted surgery on the mice to look at the liver tissue. There were very, very significant differences in the liver between the treatment and control groups, said Dr. Benny. The livers of the mice in the treatment group looked almost normal, while the livers of the control group had very large tumors. This meant that the drug could be effective against liver cancer as well.
Lastly Dr. Benny tested the mice for signs of neurotoxicity. Neurotoxicity was one of the reasons that the original TNP-470 drug had stopped advancing through clinical trials. Dr. Benny hoped that Lodamin, with its protective nanoparticles, would not have the same harmful side effects. She tested the mice for neurotoxicity by observation and experimentation. One major indicator of toxicity is weight loss, so Dr. Benny weighed the mice to determine if any mice lost weight as a result of the drug. She found no evidence of weight loss in the treated mice, so she moved on to coordination tests. Dr. Benny tested the mice in a sensitive coordination test. In this test, the mice walked on a long beam and Dr. Benny counted how many times they slipped or fell. Once again, Lodamin-treated mice did just as well as the control mice, indicating that the treated mice had no signs of neurotoxicity. These tests were done blindly, which means that the researchers performing the tests did not know which mice were in the treated group and which were in the control group. Blind tests eliminate any bias that might exist towards one group over another.
As a result of these tests, Dr. Benny concluded that Lodamin did not induce neurotoxicity in mice. Dr. Benny had succeeded in creating an orally administered drug without harmful side effects.
It is the hope of all scientists that their drugs can go to clinical trials and be available for people to use, said Dr. Benny. We are not at that stage yet, but we hope to begin clinical trials in the next few years. Dr. Benny is continuing experiments with Lodamin in animals to ensure the effectiveness and safety of the drug.
 The cover of Nature Biotechnology dedicated to Dr. Folkman's Lodamin study, as a first potent broad spectrum antiangiogenic polymeric drugThe Researchers The cover of Nature Biotechnology dedicated to Dr. Folkman's Lodamin study, as a first potent broad spectrum antiangiogenic polymeric drugThe Researchers
Sadly, Dr. Judah Folkman passed away in January, 2008, following a heart attack at the age of 74. Dr. Folkman was a Professor of Cell Biology at Harvard Medical School and the Director of the Vascular Biology Program at Childrens Hospital in Boston, MA until his death. He is remembered fondly by all. In the wake of Dr. Folkmans tragic death, Dr. Ingber of Harvard Medical School has returned to the project he helped begin many years ago.
Dr. Ingber is now the Judah Folkman Professor of Vascular
Biology in the Department of Pathology at Harvard Medical School and the Vascular
Biology Program at Childrens Hospital in Boston, MA, as well a Professor of Bioengineering at Harvard School of Engineering and Applied Sciences, and the Director of Harvards new Wyss Institute for Biologically Inspired Engineering in Cambridge, MA. He received his undergraduate, MD, and PhD degrees from Yale University, before he completed his post-doctoral fellowship under Dr. Judah Folkman at Harvard. His advice to kids is to think outside the box dont listen to what anybody tells you and to follow your passion.
Dr. Ofra Benny is a researcher in vascular biology at Harvard Medical School and Childrens Hospital in Boston. Before coming to the United States, she was a biotechnology engineer in Israel specializing in polymers and drug delivery systems to the brain. Dr. Benny has grand plans for future research and Lodamin. She is continuing experiments to learn the mechanism of Lodamin and to understand the development of metastasis in the body. Dr. Benny is also looking at the effects of Lodamin on other diseases such as macular degeneration, a disease of the eye. Of course, one of Dr. Bennys immediate goals is to begin clinical trials for Lodamin so the drug can begin to help people suffering from cancer.
To Learn More:
- Ingber D, Fujita T, Kishimoto S, Sudo K, Kanamaru
T, Brem H, Folkman J.
Synthetic analogues of fumagillin that inhibit
angiogenesis and suppress tumour growth. Nature 1990;348:555-557.
- Benny O, Fainaru O, Adini A, Cassiola F, Basinet
L, Adini I, Pravda E, Nahmias Y, Koirala S, Corfas G, DAmato RJ, Folkman
J. An orally delivered small-molecule formation with antiangiogenic
and anticancer activity. Nature Biotechnology 2008;26:799-807.
- Bourzec K. Nanotech revives a cancer drug. Available at: http://www.technologyreview.com/Nanotech/21068/page1/, Accessed October 7, 2008.
About Angiogenesis:
About Cancer:
About Nanotechnology:
Dr. Judah Folkman: In Memoriam
Written by Rebecca Kranz with Andrea
Gwosdow, Ph.D. Gwosdow
Associates
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|

Dr. Donald Ingber

Dr. Ofra Benny

Dr. Judah Folkman

Dr. Benny and a few of her colleagues
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

