| |
Highlights
The ability of a surface to absorb or repel water depends on its chemical structure. This research team developed two-dimensional (2D) nanosheets for biomedical applications using molybdenum disulfide (MoS2). By changing the atomic structure of the MoS2 nanomaterial, the researchers changed the chemical properties of the material, from water-repellant (hydrophobic) to water-absorbent (hydrophilic). This is the first demonstration that the wetting characteristic of a material can be changed by altering the atomic structure.
Water is wet. More precisely, the sensation of water or another liquid touching your skin results in what we call “wetness.” The possibility of wetness is all around us – walking in the rain or sweating, to name a few examples.
How wet a surface becomes when exposed to a liquid depends on the chemical structure of the surface. Some surfaces, like tissue or cotton cloth, readily absorb the liquid and become very wet. These are known as
hydrophilic
(literally water-loving) surfaces. Other surfaces, like finished wood or plant leaves, are water repellant or
hydrophobic.

Figure 1. A water droplet on a leaf is an example of a hydrophobic substance (photo taken by flickr user tanakawho [CC BY 2.0 (https://creativecommons.org/licenses/by/2.0)] Source: https://commons.wikimedia.org/wiki/File:Water_drop_on_a_leaf.jpg
Depending on the purpose of a material, a hydrophobic or hydrophilic surface might be more desirable. For example, a bath towel is supposed to absorb the water on your skin after you swim or bathe. It wouldn’t make sense to have a towel that simply repelled water – then the water would stay on your skin! On the other hand, many medical and electrical surfaces require hydrophobic surfaces.
Hydrophobic surfaces are typically produced by creating rougher surfaces with grooves that prevent water from penetrating the material. In many medical applications, however, the surfaces must remain smooth, so this technique of grooving the surface does not work. This is why researchers are exploring other hydrophobic materials for biomedical applications.
Dr. Akhilesh Gaharwar and colleagues at Texas A & M University are one of the research teams working to develop super-hydrophobic surfaces for biomedical applications. The focus for Dr. Gaharwar and his team is the development of two-dimensional
transition metal
dichalcogenide
nanomaterials. The material is referred to as a two-dimensional (2D) nanosheet because it is so thin – perhaps the thinnest material in the world.
The nanosheets are made up of transition metals, which are any metal found in the middle portion of the periodic table. They are typically characterized by their ability to accept electrons. Dichalcogenide refers to a class of 16 periodic elements; common elements are sulfur or selenium. These nanosheets are only several atoms thick, consisting of a single layer of metal atoms in between a sandwich of dichalcogenide atoms.
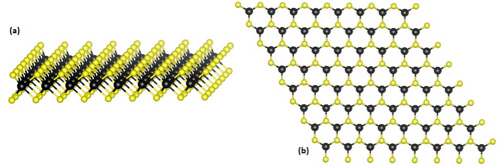
Figure 2. a) Structure of nanosheet monolayer. The transition metal is in black and the dichalcogenide elements are in yellow. b) A hexagonal nanosheet monolayer as seen from above.
(Source: https://en.wikipedia.org/wiki/Transition_metal_dichalcogenide_monolayers)
Dr. Gaharwar and his colleagues began by taking several different 2D transition metal nanosheets and dissolving them in water. The researchers wanted to study the interactions between the nanosheets and water and observe any differences between the materials.
“The difference was so clear,” recalled Dr. Gaharwar. “Molybdenum disulfide really stood out.” Out of all the materials the researchers tested, molybdenum disulfide (MoS2) was unique in that it did not dissolve in water. To better understand what was going on, the researchers used high-powered atomic microscopes to observe the atomic structure of the material. They saw a tightly packed hexagonal structure that prevented water from entering into even the first layer of atoms. That’s why the material stayed intact even when surrounded by water.
The researchers decided to see what would happen if they changed the atomic structure of the MoS2 nanomaterial. Molybdenum carries an
ionic charge
of -4 and sulfur carries a charge of +2. In the water-repellant nanomaterial, there are two sulfur atoms for every atom of molybdenum, creating a balanced, tightly knit network of atoms. The researchers added extra sulfur to the material to see what would happen. Now there were sulfur atoms that couldn’t bond to anything, creating what is known as an “atomic defect.”
When the researchers placed the nanomaterial with atomic defects in water, they saw it dissolved. They concluded that altering the chemical structure also changed the chemical properties of the material, from water-repellant (hydrophobic) to water-absorbent (hydrophilic).
Next, the researchers experimented with different ratios of molybdenum and sulfur. They calculated the angle of contact of the compounds for nanomaterials with different ratios of the two atoms ranging from 1 molybdenum: 1 sulfur to 1 molybdenum: 6 sulfur. The molecules coated a small glass plate where a water droplet was added. The material with the least sulfur (1:1 ratio) was the most hydrophobic (contact angle > 120 degrees), as indicated by the spherical shape of the water droplet. As the amount of sulfur increased in the material, the water droplet lost its shape and was eventually completely absorbed by the material (hydrophilic, contact angle close to 0). This is the first demonstration that the wetting characteristic of a material can be changed by altering the number of atomic defects.

Figure 3. Relationship between hydrophilic and hydrophobic surfaces and contact angle.
(Source: https://www.sciencedirect.com/science/article/pii/B9780323461399000086)
To further test the hydrophobic properties of the 1:1 molybdenum sulfur nanosheet, the researchers added more water droplets to the glass slide and started moving it around. They also tested different liquids like saline, blood, and vegetable oil. With each test, the compound demonstrated its super-hydrophobic properties.
Dr. Gaharwar and his team are hopeful that the molybdenum sulfur compound they have created will be able to coat a variety of surfaces to make them super-hydrophobic. This includes containers that carry blood. “Transporting blood is so important and also really complicated,” Dr. Gaharwar commented. “If a material is not super-hydrophobic, it is possible for the blood to interact with some of the components on the surface of the container.” This is why this new compound is so exciting.
For the purpose of transporting blood, Dr. Gaharwar and his colleagues went with the most hydrophobic surface they could create. A superhydrophobic surface will prevent blood clotting, which is observed while transporting blood. However, there are other situations in which the most hydrophilic surface would be ideal. For example, stem cells. Stem cells will grow and replicate when they sense a hydrophilic surface, but not a hydrophobic one. To see whether stem cells would grow on the most hydrophilic molybdenum sulfide surface, the researchers experimented with the material with six sulfur atoms to every molybdenum atom. Remember, this material had the most extra sulfur and was also the most hydrophilic.
Just as they were hoping, Dr. Gaharwar and his team found that human stem cells adhered very well to the 1:6 molybdenum sulfide compound. They hope this material will be useful for growing stem cells in the future. Other applications include scaffolding for tissue regeneration and wound healing. Dr. Gaharwar and his team are continuing to experiment with different nanomaterials to develop the best compound for each application.
Dr. Akhilesh Gaharwar is Assistant Professor of Biomedical Engineering at Texas A & M University in College Station, Texas. His research focuses on the use of nanomaterials for a variety of biomedical applications. When not in the laboratory, Dr. Gaharwar enjoys running, swimming, and spending time with his family.
For More Information:
- Jaiswal, M. et al. 2019. “Superhydrophobic states of 2D nanomaterials controlled by atomic defects can modulate cell adhesion.” Chemical Communications: 55(60): 8772-5.
- Jaiswal, M. et al. 2017. “Vacancy-driven gelation using defect-rich nanoassemblies of 2D transition metal dichalcogenides and polymeric binder for biomedical applications.” Advanced Materials. 29(36): 1702037.
- Chimene, D. et al. 2015. “Two-dimensional nanomaterials for biomedical applications: emerging trends and future prospects.” Advanced Materials, 27: 7261-84.
To Learn More:
- Gaharwar Laboratory. http://people.tamu.edu/~gaharwar/index.html
- The Water Race: Hydrophobic and Hydrophilic Surfaces. https://www.nnin.org/education-training/k-12-teachers/nanotechnology-curriculum-materials/water-race-hydrophobic-0
- Nanooze: Exploring the World of Science and Nanotechnology. http://www.nanooze.org/
- https://www.sciencedirect.com/topics/chemistry/nanosheet
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|

Dr. Akhilesh Gaharwar

Dr. Manish Jaiswal

Top row- Arnav Banerjee, Hung-Pang Lee, Kaivalya Deo, Zakary Llmao-Cohen
Row 2- Austen Kaul, Manish Jaiswal, Kanwar Abhay Singh, Giriraj Lokhande
Row 3- Kyle Robinson, Luke Conlon, David Chemene, Oscar Morales, Marcus DeAyala
Row 4- Sukanya Bhunia, Uyen Nguyen, Aparna Murli, Karli Gold, Anna Brokesh, Michael Curtis
Bottom row- Akhilesh Gaharwar, Bethel Naig, Anna Kersey
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

