Two scientists walk into a bar. A salad bar.
The scientists observe all the many types of salad greens to choose from. They are different shades of green, different shapes and sizes, different textures, and presumably they represent different flavors as well.
The scientists focus on the spinach leaves for a moment. They trace the thicker middle vein from the stem to the tip of the leaf. They notice the smaller veins branching off of the middle vein, and even smaller veins that extend from those branches. These veins reflect a larger
venous
system designed to bring nutrients from the plant stem to the furthest edges of every leaf.
Plants and animals are different in many ways, but as the two scientists at the salad bar looked at the spinach leaves, only the similarities came to mind. They recalled that humans, too, have an extensive network of blood vessels—known collectively as
vasculature—that
bring nutrients and oxygen from the heart to the extremities and back. And since these two scientists work at the intersection of
mechanical engineering
and life sciences, the plant vasculature they observed in the spinach leaves seemed to offer a potential solution to common
tissue engineering
challenges.
Tissue engineers have become very skilled at creating live tissues on a small scale. In order to scale up these tissues to be the size of functional parts of the human body, the tissues need vasculature to transport oxygen, nutrients, and waste. This is a major problem in the field of
biomedical engineering.
These scientists at the salad bar began to imagine a cross-kingdom solution—applying models from the plant kingdom to the animal kingdom to address this problem of scale.
The work that emerged from that first lunch conversation, is the topic of this month's What A Year! story. The scientists are Dr. Glenn Gaudette, Professor of Biomedical Engineering at Worcester Polytechnic Institute in Worcester, Mass., and his graduate student Joshua Gershlak. In the years since Dr. Gaudette and Josh first walked into that salad bar, they have collaborated with a wide range of researchers from plant biologists to tissue engineers that include Drs. Fabricio Medina-Bolivare and Carole L. Cramer at Arkansas State University, Dr. William L. Murphy at the University of Wisconsin at Madison, and Drs. Tanja Dominko, Pamela J. Weathers and Marsha W. Rolle at Worcester Polytechnic Institute to show that heart tissue can grow on spinach leaf vasculature with the same mechanical and electrical properties as heart tissues grown on a petri dish. The scientists hope that the next decade will see solutions to challenges ranging from repairing damaged heart tissue to
organ transplants
to
bone grafts—all
based on the spinach leaf and other members of the plant kingdom.
Spinach Scaffold
In order to make spinach leaf vasculature grow human cells, Josh first had to remove all the plant cells in a process known as
decellularization.
Josh inserted a thin plastic tube into the stem of the spinach leaf that flushed the leaf vasculature with chemical solutions to break down the plant cells. For five days, the leaves were flushed with sodium dodecyl sulfate, a basic soap that is one of the first ingredients in most body washes. The sodium dodecyl sulfate breaks up the
cell membrane
of the plants and removes all cellular material. After five days, Josh changed the chemical solution to bleach, which washed away the sodium dodecyl sulfate, sterilized the tissue, and killed any remaining
chloroplasts.
Chloroplasts are the cellular organelles that give plants their green color, so killing the chloroplasts turns the remaining spinach leaf translucent. Finally, Josh flushed water through the plant vasculature to wash away any remnants of the plant cells. The end result was a leaf-like structure with vasculature supported by the remaining plant
cellulose,
a major component of plant
cell walls.
Through decellularization, this modified spinach leaf was converted into a
scaffold—a
support structure for cells to survive and grow.

Figure 1. Decellularization of a spinach leaf
Once the spinach scaffold had been created, it was time to test its properties. To determine if the plant vasculature remained intact, Josh flushed the vasculature with a red dye. As the researchers watched the red dye trace the middle vein and then smaller veins of the spinach leaf, they knew that the vasculature had survived the decellularization process.

Figure 2. Red dye is used to confirm the plant vasculature is intact
Next, Josh tested the size of the vasculature. In order for the scaffold to be relevant for the human body, red blood cells would need to be able to fit through the veins. He flushed the veins with
microparticles
of different sizes and measured the output to determine which particles had successfully flowed through the vasculature. Since almost all of the particles the size of red blood cells were collected as output, Josh concluded that red blood cells would flow freely through the spinach scaffold.
Initial testing indicated that the spinach scaffold could support human cells, so Josh began experimenting with culturing cells both within and on top of the spinach leaf. He showed that heart muscle cells, known as
cardiomyocytes,
would grow on top of the leaf. Other cell types including stem cells also grew on the spinach leaf like they would in a petri dish. Inside the leaf, Josh injected cells that line blood vessels, known as
endothelial cells,
into the spinach veins. These attached successfully to the existing spinach vasculature.
"We were so excited that the scaffolding actually seemed to work," Josh recalled.
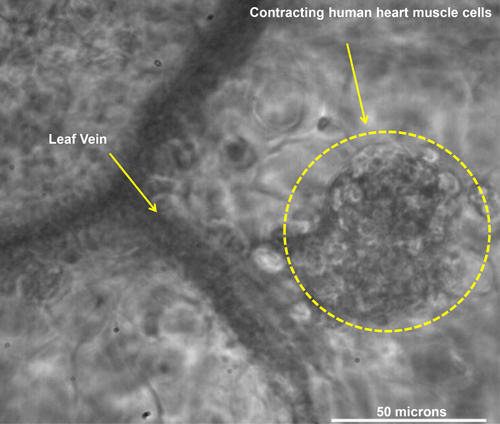
Figure 3. Cardiac muscle cells on spinach leaf
Heart cells are more complicated than many other types of cells because they function using electrical impulses to ensure that the heart contracts in the appropriate rhythm. Any engineered heart tissues, therefore, must exhibit the same electrical activity as heart cells in the body. When Josh measured the electrical activity of the cells, he found that it was indeed the same as heart cells grown in a petri dish. Josh also measured the beating of heart cells to ensure that they could contract like human heart cells do.
So far, Dr. Gaudette and Josh's research has demonstrated that spinach leaves can be decellularized and made into a scaffold to grow functional human heart cells. Importantly, this scaffold contains the vasculature to support the heart cells with oxygen and nutrients. Next, Dr. Gaudette and Josh must demonstrate the spinach scaffold is
biocompatible,
meaning that it would not be rejected if implanted into a human.
Cellulose—the compound in plant cell walls that remains after decellularization—is a commonly used
biomaterial,
and the researchers are hopeful that it will be compatible for human use. Biocompatibility tests begin in cell cultures and, if successful, move to animal models and eventually humans.
If this system is proven to be biocompatible, there is still a flow problem that the single spinach scaffold cannot address. In plants, the vasculature serves to bring nutrients and oxygen to all parts of the leaves. The flow is only one way. In humans, however, the vasculature flows both ways.
Arteries
bring oxygenated blood to the rest of the body, delivering oxygen and nutrients;
veins
bring deoxygenated blood back to the heart and lungs. In order to create a two-way system, Dr. Gaudette and Josh are working with collaborators to create a dual scaffolding with two spinach scaffolds—one leaf would serve as the arterial vasculature, bringing nutrients to the heart cells, and one would serve as the venous vasculature, bringing deoxygenated blood back to the heart.
Dr. Gaudette and Josh envision the first application of their work may be used in patients who have experienced a heart attack. During a heart attack, blood cannot flow to part or all of the heart. If a patient survives the heart attack, it is likely that some heart tissue has been destroyed due to the lack of blood flow. Damaged heart tissues cannot contract and are therefore nonfunctional. If a spinach scaffolding supporting new heart cells was introduced, however, it might be possible to re-grow the damaged heart tissue and improve the quality of life for heart attack patients.
In addition, Dr. Gaudette and Josh emphasize that there is the entire plant kingdom beyond spinach leaves to explore. For every part of the human body there could be an analogous plant system used as scaffolding. For example, a collaborator, Dr. William L. Murphy at the University of Wisconsin in
orthopedics
is currently investigating how decellularized
bamboo
stems could be used as scaffolding for bone grafts. Dandelion stems look a lot like arteries and could potentially be decellularized to replace single arteries. The plant kingdom is at least as diverse as the human body, and the possibilities are endless.
Dr. Glenn Gaudette is Professor of Biomedical Engineering at Worcester Polytechnic Institute in Worcester, MA. Dr. Gaudette began his academic career as a mechanical engineer. He entered the world of biomedical engineering by applying mechanical engineering to the human body. His research focuses on the tissue engineering of heart cells with the same mechanical and electrical properties as human heart cells to treat patients with a variety of heart conditions including heart attacks. When not in the laboratory, Dr. Gaudette enjoys watching and participating in sporting events and spending time with his family.
Joshua Gershlak is a fourth-year graduate student in Dr. Gaudette's laboratory studying biomedical engineering. His graduate work has focused on the development of tissue engineering using scaffolding based on spinach leaves. When not in the laboratory, Josh enjoys playing basketball and watching sports games.
For More Information:
- Gershlak, Joshua et al. 2017. “Crossing kingdoms: Using decellularized plants as perfusable tissue engineering scaffolds.” Biomaterials, 125: 13-22.
- Fontana, Gianluca et al. 2017. “Biofunctionalized plants as diverse biomaterials for human cell culture.” Advanced Healthcare Materials, 1601225.
To Learn More:
- Myocardial Regeneration Lab.
http://labs.wpi.edu/gaudette/
- Bioinspired Materials Lab.
http://bioinspired.engr.wisc.edu/
- Arkansas Center for Plant-Powered Production.
http://www.plantpoweredproduction.com/faculty/carole-cramer/
- National Institute for Biomedical Imaging and Bioengineering.
https://www.nibib.nih.gov/science-education/science-topics/tissue-engineering-and-regenerative-medicine
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com

