| |
Imagine youre driving along in the car and your brakes suddenly stop working properly. What would you do? If youre lucky, you would be on the highway with no one behind you, and you could just coast over to the breakdown lane. You would come to a slow, controlled stop, and make a phone call to get the car towed away and fixed. More likely, though, there will be cars all around and turns on the road, making a safe landing almost impossible. Try as you might, the car will have to either run out of gas or crash into something, whichever comes first.
Brake malfunctions are scary events and best avoided. That`s why we get our cars inspected every year. But what about our brains? In recent years, brain researchers have shown that our brains, too, can lose their brakes. When this happens, one possible outcome is the development of a brain disorder called
epilepsy.
Epilepsy is really a group of chronic brain disorders ranging from mild to debilitating. They are all characterized by recurring
seizures.
According to the Centers for Disease Control, epilepsy affects over two million adults and almost 500,000 children in the United States, with an
additional 150,000 cases diagnosed each year.
Epilepsy usually develops after a brain injury such as infection, stroke, tumor or traumatic injury. Symptoms of epilepsy, however, may not appear until weeks, months or even years after the initial traumatic event.
In February 2007, we profiled
Dr. Shelley Russeck
of Boston University School of Medicine who was conducting research on a type of epilepsy called
Temporal Lobe Epilepsy.
Dr. Russeck and her colleagues had narrowed in on a single unit of a specific signaling molecule released by interneurons called Gamma-Amino Butyric Acid, or GABA for short.
In some cases of epilepsy, so much time has passed since the initial brain injury that it is unclear for some patients why the condition has developed at all.
This month, Drs. Chris Dulla and David Cantu at Tufts Medical School in Boston, MA have built on the research of Dr. Russeck and other colleagues working on GABA, epilepsy, and other
neurological disorders.
In some cases of epilepsy, so much time has passed since the initial brain injury that it is unclear for some patients why the condition has developed at all. Drs. Dulla and Cantu wanted to better understand what changes were taking place in the brain during this
latent period.
We dont notice behavioral changes in the patient during this time, explains Dr. Dulla. But we suspected that within the brain there was a lot going on to trigger the development of epilepsy, and we wanted to try to figure that out. In other words, they wanted to find out what had happened to the brain`s braking mechanisms.
In a healthy brain, brain activity is tightly regulated by
neurons
that either promote or prevent continued neuron activity. Neurons that promote brain activity are known as
excitatory neurons;
those that prevent brain activity are called
inhibitory neurons.
Neurons that connect other neurons are called
interneurons.
Interneurons usually relay inhibitory messages from other neurons. In a healthy brain, excitatory and inhibitory neurons work together to regulate brain activity. In an epileptic brain, however, Drs. Dulla and Cantu observed a marked decrease in inhibitory interneurons. This means that the epileptic brain has lost its brakes, commented Dr. Cantu. It has no way to slow down and inhibit brain activity. As a result, the brain is overactive. Eventually this overactive brain crashes, resulting in an array of symptoms that characterize epileptic seizures such as sensory disruption, convulsions and unconsciousness.
To conduct their research, Drs. Dulla and Cantu used a variety of techniques to measure brain activity in mice that experienced a brain injury. First, the researchers used
electrophysiology
to measure the electrical activity of slices of mice brains. Next, they conducted
immunohistochemistry assays,
which enabled them to stain and visualize the particular neurons of interest. This allowed the researchers to quantify exactly how many neurons had been lost during the latent period. By comparing these results with those of controls, these researchers confirmed the loss of inhibitory interneurons after brain injury in mice.
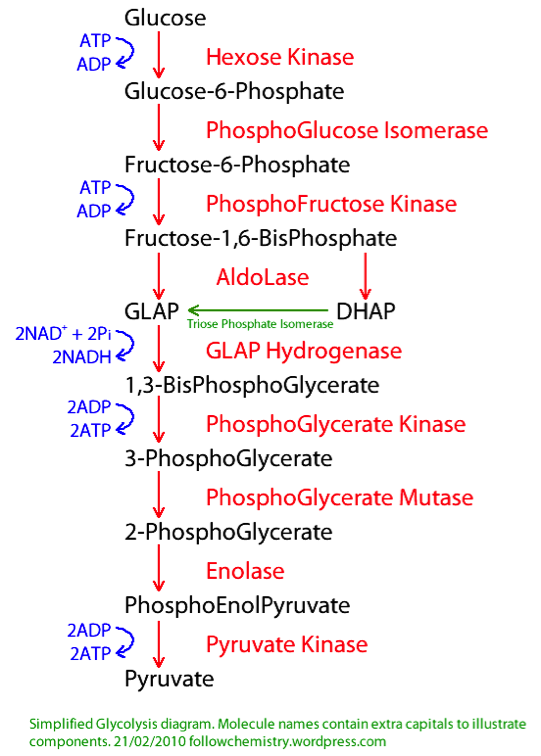
Glycolysis
Drs. Dulla and Cantu hypothesized that preventing the process of glycolysis in the post-injury brain could reduce the over-activity they had observed. Do you remember studying the process of
glycolysis
in your biology classes? (If you havent yet, youll get there soon!) Glycolysis is one of the most basic cellular processes that transforms
glucose
into energy
(adenosine triphosphate,
or ATP). It is an integral part of all introductory biology courses because it is the basis of cellular function. Do you remember having to memorize all the steps involved in glycolysis? I do! jokes Dr. Cantu. And now Im working on a real-life application!
Eventually this overactive brain crashes, resulting in an array of symptoms that characterize epileptic seizures such as sensory disruption, convulsions and unconsciousness.
To test their glycolysis hypothesis, these researchers used a molecule called 2-deoxy-glucose (2-DG), which is known to inhibit the
enzyme
hexokinase.
Hexokinase is the very first enzyme necessary for glycolysis to even begin. By using 2-deoxy-glucose, the researchers intended to stop glycolysisand therefore decrease brain activityin the overactive brain. The molecule 2-deoxy-glucose is very similar to glucose, so it can easily enter cells.
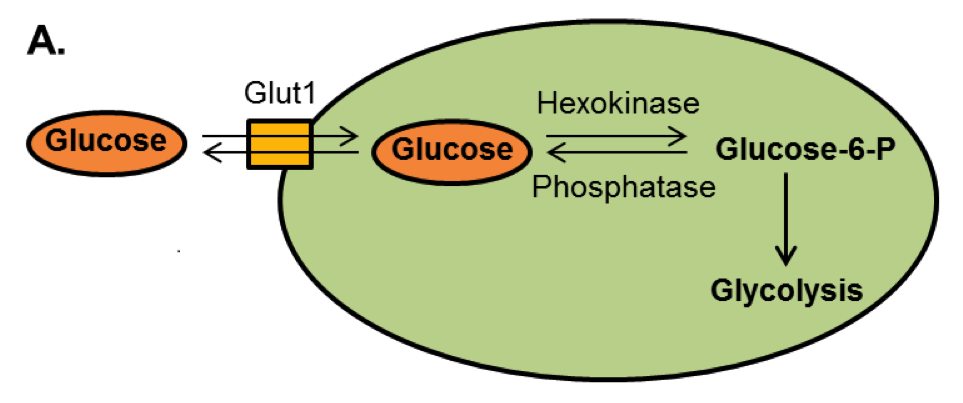
Glycolysis begins with glucose entering the cell and its conversion to glucose-6-phosphate.
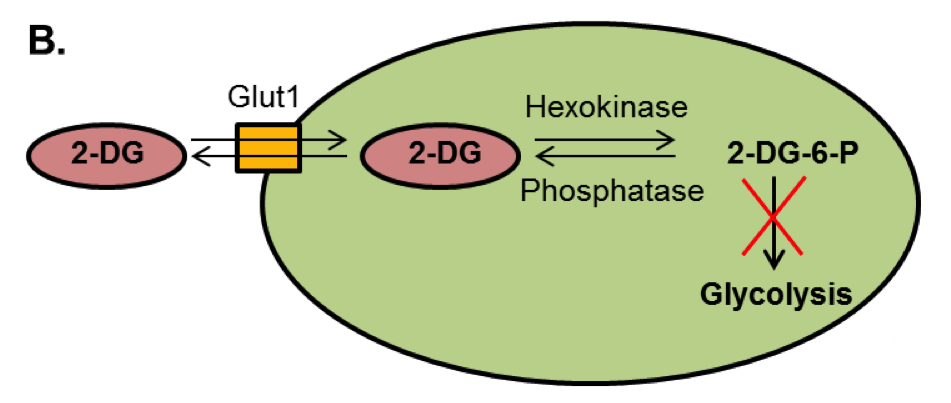
2-DG blocks glycolysis by being converted to 2-DG-6 phosphate.
In this next set of experiments, the researchers focused on injured brain slices. Untreated injured brain slices have a very high level of electrical activity. When exposed to 2-deoxy-glucose, however, these same brain cells went back to a normal level of electrical activity in just 30 minutes. With these positive findings, the researchers then moved to mouse models, where they treated animals that had experienced brain injury with 2-deoxy-glucose for one week following the initial traumatic event. We found the electrical activity significantly decreased in animals treated with 2-deoxy-glucose, explains Dr. Cantu. Drs. Cantu and Dulla suspect that the changes in electrical activity they have observed are due to a reduced loss of interneurons. But they arent sure yet. Thats a hypothesis were currently testing, confirms Dr. Dulla.
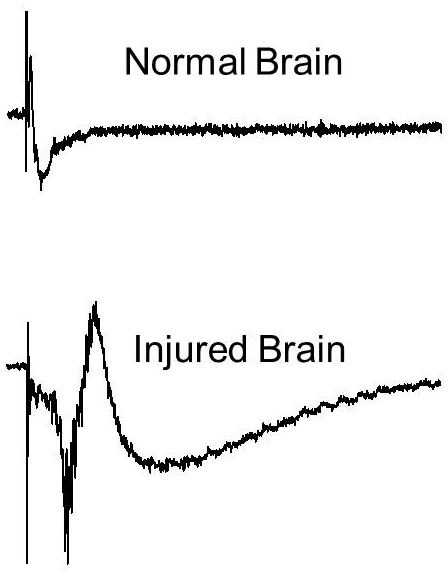
Recordings of nerve cells from normal and injured brains.
If the researchers can figure out what has happened to the brakes and therefore what makes the brain overactive, it opens all sorts of possibilities to try to fix them. Drs. Dulla and Cantu hope one day they will have treatment options to help restore typical brain activity.
Thats what keeps us motivated, exclaims Dr. Cantu. We love doing science, but also know our findings could really affect peoples lives.
In addition, interneurons are involved in many other types of neurological diseases ranging from
autism
to
Alzheimers disease,
so the results of this research could have widespread implications.
In future research, Dr. Cantu plans to study different
metabolic pathways
and target additional enzymes involved in glycolysis in the hope that metabolic inhibition can decrease the incidence of seizure in the epileptic brain. Dr. Dullas future work will focus on how groups of neurons work together. He hopes to identify other types of neurons that are lost or preserved during brain injury. This will enable researchers to understand how neurons that stay alive after brain injury manage to do so, with the hope of preserving more neurons after an initial traumatic event.
Dr. Chris Dulla is Assistant Professor of Neuroscience at Tufts University School of Medicine. His research focuses on how different neurons work together and how their functions differ in the healthy and damaged brain. He is also interested in developing new and better ways to visualize the brain and brain activity. When not in the laboratory, Dr. Dulla enjoys spending time with his family, playing guitar, camping and surfing.
Dr. David Cantu is a post-doctoral fellow in the laboratory of Dr. Chris Dulla at Tufts University School of Medicine. His research interests include cellular metabolism in the brain and how metabolism is affected by neurological disorders. In addition to his research, Dr. Cantu is a recipient of the Institutional Research and Academic Career Development Award from the National Institutes of Health. This award enables Dr. Cantu to teach undergraduate classes at partnership institutions. This past semester, he taught a class in cell biology at Bunker Hill Community College in Boston, MA, one of Tufts partner institutions.
To Learn More:
- Cantu, D. et al. 2014. Traumatic Brain Injury Increases Corticol Glutamate Network Activity by Compromising GABAergic Control. Cerebral Cortex 2014 Mar 7. [Epub ahead of print]
For More Information:
Epilepsy
- Epilepsy Foundation. www.epilepsy.com
- National Institutes of Neurological Disorders and Stroke. www.ninds.nih.gov/disorders/epilepsy/epilepsy.htm
- World Health Organization. www.who.int/topics/epilepsy/en
Other Neurological Disorders
- National Institute of Neurological Disorders and Stroke. www.ninds.nih.gov/disorders/epilepsy/epilepsy.htm
- American Academy of Neurology. patients.aan.com/disorders
- World Health Organization. www.who.int/features/qa/55/en
- Scientific American. www.scientificamerican.com/neurological-disorders
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|

Dr. Chris Dulla

Dr. David Cantu

Postdoctoral Scholar Kendall Walker

Doctoral candidate Lauren Andresen

Associate Professor Giuseppina Tesco

UK age-band statistics on epilepsy

US projected prevalence of epilepsy
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

