| |
Did you know that there is a parasite that infects nearly one-third of the worlds human population? The parasite, Toxoplasma gondii, affects almost one quarter of the U.S. population over the age of 12 (Centers for Disease Control and Prevention). But there are some regions of the world where 95 percent of the population is infected throughout most of their lives (Centers for Disease Control).
T. gondii causes a disease called toxoplasmosis. Why have you never heard of the parasite T. gondii and toxoplasmosis? One major reason is that most people who are infected with the parasite do not have obvious symptoms. And when symptoms of a new infection do occur, they are usually flu-like and are often misdiagnosed. In any event, the symptoms of the acute infection generally clear up within several weeks, without the infected persons even knowing what caused them. Although healthy adults can generally ward off the infection without severe symptoms, the results of toxoplasmosis can be much more dangerous for children and people with compromised immune systems.
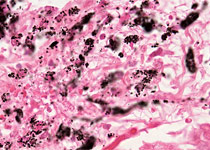
Toxoplasmosis
(from the Human Pathology Digital Image Gallery)
There are three main ways T. gondii can be transmitted. First, you can ingest the parasite T. gondii by eating raw or undercooked meats or unwashed fruits and vegetables. Second, T. gondii can be acquired from materials contaminated by cat feces. The cat intestine provides a unique environment for a highly infectious form of the parasite to reproduce. The oocysts that result are present in cat feces and one cat can excrete 20 million oocysts in a period of approximately 20 days. People contract the infection by coming into contact with the oocysts. This can happen when you change a cats litter box or if you garden in or eat produce from a garden where a cat has defecated, or play in a sandbox where a cat has defecated. Third, the infection can be passed from mother to child if the mother is infected by the parasite for the first time while she is pregnant.
Healthy adults who contract the infection through produce, meats, or contact with cat-excreted oocysts may not have any obvious symptoms. If an immunosuppressed person becomes infected, though, the infection may cause headaches, seizures, nausea, and eye disease in severe cases. Severe symptoms can also occur when the infection is passed congenitally from the acutely infected or immune-compromised mother to her unborn baby. Congenital infection of the fetus may result in a miscarriage or stillborn infant. Children who survive the infection occurring late during their time in the womb often look normal but may develop mental disabilities and eye disease later on in life. Unfortunately, only two states require testing of newborns for toxoplasmosis. It is estimated that one in every five thousand children is born with toxoplasmosis in the United States.
Current drugs to treat toxoplasmosis have potentially serious side effects, making their use problematic. There is, however, a potential new drug for toxoplasmosis that will soon enter clinical trials. This is the result of the collaborations of Dr. Rima McLeod from the University of Chicago Medical Center with Drs. David Jacobus and Wil Milhous, both formerly of Walter Reed Army Institute of Research in Silver Spring, Maryland.
Malaria
Dr. McLeod knew of the similarities between the T. gondii parasites and the parasites that cause malaria called Plasmodia. Malaria is a much more widely known infection because of its high mortality rate. Mosquitoes carry Plasmodial parasites in the same way that cats carry the T. gondii parasite. There are several anti-malarial drugs available, but problems with development of drug resistance limit their use. All of the current anti-malarial drugs stem from the work of Drs. Jacobus and Millhous, who have devoted their lives to developing drugs to combat malaria, and have saved hundreds of thousands of lives by this work.
Because of the close link between malaria and toxoplasmosis, Dr. McLeod wanted to test anti-malarial drugs to determine their effects on toxoplasmosis. In order to do so, she collaborated with Dr. Jacobus (now of Jacobus Pharmaceuticals), Dr. Millhous (now of University of Southern Florida), and others at the Walter Reed Army Institute of Research.
The anti-malarial drugs are engineered to target certain organelles that occur in the parasites but not in the human, or they can target enzymes that are structurally different in parasites and humans. The most recent anti-malarial drugs target an enzyme called dihydrofolate reductase, (thankfully shortened to DHFR). Parasites use DHFR to synthesize a chemical called that is important to their survival. A new anti-malarial drug called JPC-2067B is much better than previous drugs because the parasites do not develop resistance to it.
A Note on Drug Resistance
When you take an anti-parasite medicine, such as the anti-malarial drug, it kills most (perhaps 99.9 %) of the parasite, but some (perhaps 0.1%) of the parasites survive. The surviving parasites can sometimes prosper because they have resistance to the drug and if they replicate, they can make additional drug-resistant offspring. Drug resistance to each anti-malarial drug has developed about every ten years. However, this new anti-malarial drug (JPC-2067B) binds so tightly to the DHFR that scientists predict that resistance may not develop. Researchers plan to use the new drug in combination with another highly effective medicine. Such a combination of two drugs will further diminish the likelihood resistance will develop.
Dr. McLeod wanted to test this new drug in treating T. gondii infections as well. To study its effectiveness on toxoplasmosis, Dr. McLeod and her colleague Ernest Mui first used a tissue culture system containing the T. gondii parasite. [Tissue cultures are grown in vitro(= in glass), meaning, in a test tube, Petri dish or multi-well tissue tray, as opposed to in vivo (= in a living body).] They treated half of the cultures with the drug, while the others were controls. Dr. McLeod and Mr. Mui found a significant decrease in the amount of T. gondii growing in cultures treated with JPC-2067B compared to the controls. Specifically, when they quantified the results, they found 10-fold less T. gondii in the treated cultures compared to the untreated cultures.
This was a promising finding that demonstrated the effectiveness of this new anti-malarial drug (JPC-2067B) on T. gondii. Then, to be sure that the drug worked on the T. gondii parasite, Dr. McLeod and Mr. Mui repeated these experiments. In every case they found the tissue cultures treated with JPC-2067B significantly inhibited parasite growth compared to untreated controls. When compared to the current drug used to treat toxoplasmosis, JPC-2067B worked much better. It killed more parasites with ten-fold lower doses of the drug needed to produce this effect.
The next step was to test the effectiveness of JPC-2067B in mice. Dr. McLeod and Mr. Mui inoculated laboratory mice with the T. gondii parasite. They then gave half of the mice the JPC-2067B. The other half made up the control group. Dr. McLeod found that the mice treated with JPC-2067B had fewer parasites in their bodies compared to the control group. These experiments showed JPC-2067B was effective against the T. gondii parasite in mice. Additional studies showed the drug was safe and did not cause any harm to mice or monkeys. In addition to its effects on T. gondii, the drug is effective against the DHFR enzyme from malaria and other opportunistic microorganisms.
The next step will be to test the effectiveness of JPC-2067B against T. gondii in humans. It is hoped that clinical trials will begin within the year. These results are promising for the effectiveness of anti-malarial drugs against T. gondii in humans, said Dr. McLeod. They provide hope for the millions of people in the United States and around the world who are infected with toxoplasmosis.
Dr. Rima McLeod is research scientist and a physician who specializes in infectious diseases.
She is a Professor of Ophthalmology & Visual Science, Medicine, and Pathology at the University of Chicago and the Medical Director of the Toxoplasmosis Center. Dr. McLeods research centers on the study of toxoplasmosis and the parasite T. gondii. Her projects include creating new drugs to treat toxoplasmosis and also creating a vaccine that could be used to protect against the infection. Dr. McLeod is also involved in public awareness campaigns. Currently Massachusetts and New Hampshire are the only states to test for T. gondii in newborns. Dr. McLeod would like to see this test mandatory in all hospitals in every state.
To Learn More:
- Mui, Ernest et al. Novel Triazine JPC-2067-B Inhibits Toxoplasmosa gondii In Vitro and In Vivo. Neglected Tropical Diseases, 2008(2): e190.
For More Information:
Written by Rebecca Kranz with Andrea
Gwosdow, Ph.D. Gwosdow
Associates
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|
 Dr. Rima McLeod Dr. Rima McLeod
 Members of the McLeod Lab Members of the McLeod Lab
 Members of the McLeod Research Group Members of the McLeod Research Group
 Bacterial Drug Resistance Bacterial Drug Resistance
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

