
Figure 1: Dr. Carlos Chagas
For millennia humans have desired to evade death through eternal life. Both the Sumerian hero
Gilgamesh
and the Spanish conquistador
Juan Ponce de Leon
went on long quests to prolong their lives indefinitely and achieve immortality.
Yet death is a necessary and natural process, for the whole organism and for individual cells. All human cells have the ability to perform apoptosis, or cell death. It is critical for sick, infected or unproductive cells to make room for healthier ones so that our bodies can continue to function. What would happen if cells were to stop the natural apoptotic processes and continue to survive beyond the bodys need for them? New research into the life cycle of the parasite that causes
Chagas disease begins to answer this question. In 1909, Brazilian doctor Carlos Chagas identified the parasite Trypanosoma cruzi as the cause of the infection called Chagas disease. T. cruzi parasites are found in the feces of some
reduviid bugs. In the language of infection, the bugs are the vector of the disease
that is, they deliver the parasite from an infected animal or person to another, non-infected animal or person.
The reduviid bugs are commonly known as kissing bugs for their tendency to bite around the eyes and mouth at night. The bite does not hurt, so most people dont realize they have been bitten. The T. cruzi parasite, which is left behind in the bugs feces, then migrates to the mouth, nose, or eye cavities on the face, where the infection begins. If T. cruzi are deposited far from these openings, it can still gain entry if there is a cut or abrasion. Importantly, though, T. cruzi cannot penetrate the skin by itself.
Two Decades Must Seem Like Eternity to a Parasite
After the initial infection of T. cruzi, a small number of people will immediately develop symptoms known as acute Chagas infection. Acute Chagas infection can be diagnosed through a blood test: treatments are available, although the treatments do not completely eliminate the parasites from the body. Most people will not develop symptoms and will never know they have been infected. That is because the parasites remain in the bloodstream for a few months before disappearing into the tissue where they remain indefinitely. So a person infected with T. cruzi might never show symptoms, or might show them 20 years later. The number of people with latent T. cruzi infection is unknown because the parasite cannot be detected once it enters the tissue.
“We asked ourselves, How is it possible that the host cells stay alive for so long with thousands of T. cruzi parasites consuming the host cells vital resources? We discovered that PDNF on the surface of the T. cruzi parasite essentially inhibits cell death signals and activates cell-protective mechanisms, ensuring T. cruzi sufficient time to develop and reproduce in the host cell.”
In approximately 30% of cases patients will develop the symptoms of Chagas disease several decades later. Chagas disease affects over 10 million people in the Americas including the United States. It most commonly presents itself as an enlarged heart that can lead to a heart attack or heart rhythm abnormalities that cause sudden deaths. Many patients will also report difficult or painful swallowing and defecating as a result of an enlarged gut. Although it is possible to treat these symptoms, there is no known cure for Chagas disease.
Mercio Perrin, MD, PhD, Professor of
Pathology at Tufts University School of Medicine, has spent his career studying the T. cruzi parasite that causes Chagas disease. Specifically, Dr. Perrin is fascinated by the ability of T. cruzi parasite to remain inside tissue cells for so long, 20 years or more.
What is a Receptor?
A receptor is a protein on the surface of the cell that binds to specific molecules outside the cell and activates molecules within the cell. Different receptors bind to different molecules outside the cell in order to start different processes inside the cell.
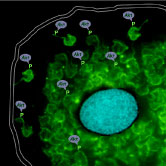
A human cell infected with Trypanosoma cruzi. Akt kinase (shown in purple) activates PDNF (shown in green) on the parasite surface.
The sneaky mechanism by which T. cruzi parasite enters the host cell has been known for many years. T. cruzi produces a protein called PDNF that mimics
neurotrophic factors produced by humans, so it can bind to the same receptors. Specifically, PDNF is able to bind to sialic acid. Sialic acid is a common surface molecule, but it is not present within the cytosol, the fluid inside the cell, where T. cruzi spends most of its life. This presented Dr. Perrin with a puzzle. If T. cruzi spends most of its life cycle inside the cell, why would it bind to something only present on the cells outside surface?
In order to answer this question, Dr. Perrin and Marina Chuenkova, PhD, a research instructor in the pathology department at Tufts School of Medicine, used a computer model that compares
amino acid sequences of proteins and related them to the known functions of other proteins. Using this analysis, Dr. Perrin noticed that PDNF contained sequences that were important for the reaction of specific
enzymes
within the cell. When these enzymes activate, they promote survival and prevent cell death. Dr. Perrin hypothesized that T. cruzi made PDNF to bind to these specific enzymes and not to bind to sialic acid.
The Experiments
To test this hypothesis, Drs. Perrin and Chuenkova performed several
in vitro experiments with T. cruzi and PDNF. In one set of experiments Drs. Perrin and Chuenkova cultured cells that would produce PDNF and then treated those cells with hydrogen peroxide, a toxin that will normally kill cells. The scientists predicted that if PDNF were inside the cell, it would trigger survival mechanisms so that the cell would resist chemicals that would otherwise kill it. Indeed, this is what Drs. Perrin and Chuenkova found. Next, they tested cells infected with the T. cruzi parasites that produce PDNF. These cells also resisted the hydrogen peroxide toxin. As a control, Drs. Perrin and Chuenkova treated normal cells with hydrogen peroxide and all of the normal cells died, as expected. From these experiments, they concluded that T. cruzi makes cells resistant to outside regulation through the production of PDNF.
Dr. Perrin believes that T. cruzi can survive inside the host cell for so long because it activates cell survival mechanisms from both the inside and the outside of the cell. In order for T. cruzi to enter the cell, PDNF binds to a particular surface receptor known as the
Trk (pronounced track) receptor. Not only does this allow T. cruzi to enter into the cell, the activation of Trk also activates the enzymes within the cell that inhibit cell death. Once inside, T. cruzi binds to these enzymes so they remain active. Dr. Perrin has also shown that the T. cruzi parasite inhibits the cell division process. This is an incredibly novel adaptation by a parasite, remarked Dr. Perrin. Many parasites bind to surface molecules to gain entry into the cell and in the process of binding, activate a chain reaction inside the host cells, but we have never seen a case where the parasite also directly binds and activates the same intracellular molecules normally activated from the outside of the cells, explained Dr. Perrin.
“In short, the T. cruzi parasite has a means of establishing life insurance once it has invaded the host. If we can fully understand the mechanisms behind this protection, we can begin to explore ways to undermine it with treatment.”
Future Work and Possible Benefits
With this information, Dr. Perrin hopes treatments will be developed that prevent Chagas disease by blocking the interaction of PDNF with both the cell surface receptor and intracellular enzymes. The goal is to prevent the PDNF carried by T. cruzi from binding to the Trk receptors on the outside of the cell. One method for doing this is to produce a synthetic PDNF that will bind to the Trk receptors first, before the T. cruzi can produce PDNF. Dr. Perrin and his colleagues have shown that this occurs through in vitro experiments. Dr. Perrin took a tissue culture and added large amounts of PDNF. The PDNF bound to the Trk receptor, preventing the T. cruzi parasites from doing so. Another method Dr. Perrin is exploring is the development of an antibody that would bind to the Trk receptor in a similar way.
In the future, Dr. Perrin also hopes to study the mechanisms that allow T. cruzi to inhibit cell death so that they can be applied to other diseases in which this would be a desirable outcome. For example, there are many diseases where tissues degenerate for genetic or other unknown reasons and the cells die. Dr. Perrin hopes to one day be able to use his T. cruzi and PDNF research to give cells the ability to fight these deadly diseases unrelated to Chagas disease.
Dr. Mercio Perrin is a professor in the pathology department at Tufts University School of Medicine and member of the immunology program faculty at the Sackler School of Graduate Biomedical Sciences at Tufts. He studies the parasite Trypanasoma cruzi that causes Chagas disease. Dr. Perrin was born in Brazil and received his medical degree there. He worked as a doctor in Brazil where he encountered many patients infected with Chagas disease. After several years in practice, Dr. Perrin came to New York for graduate school where he specialized in carbohydrate-protein interactions. It is in this context that he discovered PDNF in sialic acid and began working with the T. cruzi parasite. This has been the focus of his research ever since. Outside of the lab, Dr. Perrin enjoys biking and playing soccer.
For more information:
- Chuenkova, M. and M. Perrin. (2009). “Trypanosoma cruzi Targets Akt in Host Cells as an Intracellular Antiapoptotic Strategy.” Science Signaling, 2(97).
To Learn More:
- Centers for Disease Control and Prevention: www.cdc.gov/chagas
- World Health Organization. www.who.int/topics/chagas_disease/en/
- Chagas Disease Foundation. www.chagasfound.org/
- University of Texas, Arlington.
www.uta.edu/chagas
Rebecca Kranz with Andrea
Gwosdow, Ph.D. Gwosdow
Associates
Image Credits
Figure 1, Dr. Carlos Chagas -
http://popularizacaodaciencianaescol.blogspot.com
Gilgamesh, King of Uruk - http://electricliterature.com/blog/2010/02/10/gilgamesh-the-videogame-review
Juan Ponce de León y Figueroa - http://ushistoryimages.com/juan-ponce-de-leon.shtm
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |

