| |
Tissue regeneration is the process of growing new tissues. In nature, limbs are the most common tissue to regenerate. Amphibia, such as salamanders, are known to regrow limbs. In humans, the tips of fingers have been known to regrow.
In some of the first experiments with tissue regeneration in humans, scientists would inject new cells into the body to see what would happen. They generally didn't find very much. The cells would migrate away from the injection site and the researchers wouldn't know where they went.
About twenty years ago, the Vacanti brothers—Charles, an anesthesiologist and Joseph, a pediatric surgeon—developed a novel technique for introducing new cells into the body. Rather than inject the cells directly, they attached the cells to an implantable support called a scaffold. This scaffolding was made from different types of plastic and was designed to biodegrade after a certain amount of time. The researchers found that cells attached to this type of scaffolding did not migrate away from the site of implantation like previously injected cells. Furthermore, oxygen actually diffused across the scaffolding and kept the cells alive.
To test this design, the researchers implanted cells attached to the scaffolding into a mouse. For their first experiments, they chose the tissue called cartilage, found in your ears, nose, and most of your joints. Cartilage was a good tissue type for these experiments because there is only one type of cell — chondrocyte — and very little blood supply is needed to provide the cells with oxygen.
The researchers found that cartilage tissue attached to the scaffolding and implanted into a mouse model did, in fact, grow. This can be seen in the famous mouse model with cartilage tissue grown in the shape of a human ear.
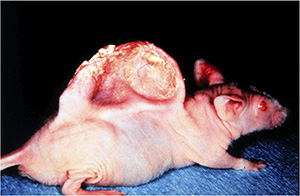
The results from the cartilage experiments provided further evidence that this was a viable method for tissue regeneration. Dr. Charles Vacanti, the anesthesiologist, decided to pursue this line of research with spinal cord injuries. He chose spinal cord injuries because they are the most devastating in the body, and they're often caused by very minor damage: just a few millimeters of damaged tissue can leave you paralyzed.
Spinal Cord
There are many different types of cells in the spinal cord. Rather than attempt to grow them on the scaffold, Dr. Vacanti used immature stem cells, which have the ability to become any type of spinal cord cell. He placed stem cells on the scaffold and introduced them into the body. To test the effectiveness of the spinal cord scaffold, the researchers first observed the difference between spinal cord cells grown in the laboratory with and without the scaffolding. They found that both groups of cells mimicked spinal cord cell growth in animals, confirming that the scaffolding itself did not affect cell growth.
Next, the researchers tested their model in rats. To do this, they implanted the device into rats with small spinal cord injuries. These rats were injured to the extent that they had very little muscle control. After implantation, the researchers observed the animals over the course of several months. The researchers observed increased muscle function capability in the rats. These results are very promising for Dr. Vacanti's scaffold model, tissue regeneration techniques, and of course, in the future, for patients with spinal cord injuries.
How the scaffold may work
Dr. Vacanti hypothesizes that the scaffold works to allow neurons to build new connections after injury. This is normally what damaged nerve tissue would want to do, but often there is too much scar tissue, which disrupts the connections. The scaffolding provides support for these new bridges to be built, allowing for increased motor function in the mice. Dr. Vacanti suspects that simply removing scar tissue after spinal cord injury might significantly increase the prognosis for patients. In his next set of experiments, he would like to test this hypothesis in monkeys and in humans who have suffered a spinal cord injury. If these experiments prove successful, he hopes to introduce the spinal cord scaffolding to further improve recovery and function.
Dr. Charles Vacanti is a Professor of Anesthesiology at Harvard Medical School. He works as a clinical anesthesiologist in the operating room and also conducts research on tissue regeneration. When not in the lab, Dr. Vacanti has many hobbies, including collecting and repairing antique motorcycles.
For More Information:
- Zhou, L. et al. 2011. "Engineering ear constructs with a composite scaffold to maintain dimensions." Tissue Engineering Part A, 17(11-12): 1573-81.
- Haisch, A. 2010. "Ear reconstruction through tissue engineering." Advances in otorhinolaryngology, 68: 108-119.
- Vacanti, M. 2008. "Composite tissue formation derived solely from a blood biological matrix: a preliminary study." 40(5); 1696-9.
- Vacanti, C. 2006. "The history of tissue engineering." Journal of Cellular and Molecular Medicine, 10(3): 569-576.
To Learn More:
- Tabesh, H. et al. 2009. "The role of biodegradable engineered scaffolds seeded with Schwann cells for spinal cord regeneration." Neurochemistry International, 54(2): 73-83.
- Spinal cord trauma http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0002061/
- Spinal cord injuries http://www.nlm.nih.gov/medlineplus/spinalcordinjuries.html
- National Spinal Cord Injury Association http://www.spinalcord.org/
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|
Read an article about the four Vacanti brothers
Three Interesting Videos About Tissue Regeneration
Growing implant tissue on 3-D scaffolds

Tissue Engineering Scaffold Visualization

Regenerative Medicine: Re-Growing Body Parts
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

