| |
Have you ever wondered how you can see? Sight is an amazing tool critical to human survival. We depend on our eyes to capture images and then send those images to the brain so it can interpret them and give meaning to the world around us. You can think of the eyes as the data collectors and the brain as the data analyzer for your vision.
What we generally think of as the eye, in humans that strange shape sort of like an oval, is really just a small portion of the eye. If you feel around your eye socket youll find that the eye is actually shaped like a ball. The adult eyeball is about one inch in diameter. 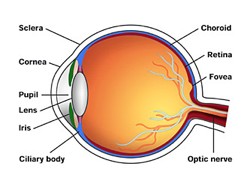 Image from http://www.healthyeyes.org.uk/uploads/pics/Eye-1.jpg Image from http://www.healthyeyes.org.uk/uploads/pics/Eye-1.jpg
The eyeball is filled by a gelatinous core the vitreous and surrounded by three layers that serve to protect it and to gather visual information. (Refer to the picture on the right.) You may have heard of the sclera and the cornea. The sclera is the white tissue protecting the eyeball and the cornea is a clear window inserted into the sclera that allows the light to enter the eyeball, but still protect it. Moreover the middle layer contains some muscle fibers, supporting cells and the choroid which is a layer of capillaries that provides oxygen and nourishment to the nerve tissue called the retina. The middle layer also contains the iris, which adjusts the amount of light entering through the pupil like a shutter of a camera. The iris gives your eyes their color.
The lens is right behind the iris: it focuses the light at the back of the eye onto the retina. The retina is the innermost layer of the eyeball. It is made up of different types of neurons (specialized nerves) that receive light and send signals to the brain. The three different categories of neurons are: photoreceptor neurons that receive the light, interneurons (bipolar, amacrine and horizontal cell) that process the information to retinal ganglion cells (RGC). The axons of the retinal ganglion cells are connected to the brain by the optic nerve by which the retina sends visual signals to the brain.
As you can see, though it is a small organ, the eye is not a simple one. In fact, it is so complex that some ophthalmologists (eye doctors) specialize in one small portion of the eye, the back of the eye or the front of the eye, for example.
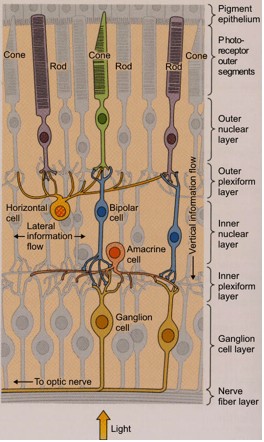 http://www.fz-juelich.de/inb/inb-1/Photoreception/ There are two main types of photoreceptors in the retina, called rods and cones, named for the different cell shapes. They absorb photons (light) and convert the light signal into a biochemical signal and then send it into an electrical signal to the interneurons, bipolar and amacrine cells. The interneurons then pass the signal along to the ganglion cells. All the ganglion cell axons together form the optic nerve. Signals are sent through the optic nerve to the visual cortex in the brain where the signals are interpreted. (By the way, all of this happens very, very quickly.) Take a look at the diagram to the left to see the path of light from the retina to the brain. http://www.fz-juelich.de/inb/inb-1/Photoreception/ There are two main types of photoreceptors in the retina, called rods and cones, named for the different cell shapes. They absorb photons (light) and convert the light signal into a biochemical signal and then send it into an electrical signal to the interneurons, bipolar and amacrine cells. The interneurons then pass the signal along to the ganglion cells. All the ganglion cell axons together form the optic nerve. Signals are sent through the optic nerve to the visual cortex in the brain where the signals are interpreted. (By the way, all of this happens very, very quickly.) Take a look at the diagram to the left to see the path of light from the retina to the brain.
Glial Cells: Worth Paying Attention To
The neurons in the retina, like all neurons, are supported by glial cells that provide structure to the neurons and are vital to proper neuron function, and participate in signal transmission in the retina. The particular glial cells found in the retina are known as Müller glial cells. Glial cells are important to the research in this story.
Humans are Mammals
It is important to know that neurons in the mammalian retina do not regenerate after a certain point in embryonic development. You are born with all the retina neurons youre ever going to have. This means that when retinal neurons are lost, the ability to see is also lost. Blindness can occur even when only one particular type of neuron is lost. For example, the eye disease called macular degeneration is caused by the loss of photoreceptor cells, while the eye disease called glaucoma is caused by the loss of ganglion cells. If there was a way to stimulate retinal cells to regenerate, though, it could lead to possible treatments and cures for these and other retinal neurodegenerative diseases.
The ability to regenerate body parts is seen throughout the animal world, but not so much in humans. Glass lizards can regrow their tails if bitten off by a predator and starfish can regrow severed limbs. It has been known since the 1700s that amphibians have the ability to regenerate their retinas when damaged. Researchers have marveled and studied this feat for centuries in hopes of understanding and harnessing the power of retinal regeneration. Some scientists found that they could remove the entire retina from a salamander and the salamander would replace it entirely.
Dr. Thomas Reh of the University of Washington School of Medicine was one such student who was amazed to learn of the regenerating power of amphibians. It posed the question: why cant we do that? remarked Dr. Reh. He decided to study retinal regeneration in the hopes of understanding the mechanism of neuronal regeneration and applying that information to regenerate retinal cells in humans. Dr. Reh began his work in graduate school with hamsters, looking at the regenerating power of newly-born animals. As a post-doctoral fellow, Dr. Reh and other researchers also investigated this process in fish and frogs. Fish and amphibians repair their retinas very well, but it was believed for many years that birds lacked this ability said Dr. Reh. However, after setting up his own lab, working now with postdoctoral fellows, he found that if the retina was damaged in newly hatched chicks, they are able to regenerate some of the damaged retinal neurons. After we discovered that birds could make new retinal neurons after damage, we started to wonder about mice.
Mice are mammals, like humans, and closer to humans evolutionarily than either fish or amphibians. Dr. Reh and his research fellow, Dr. Mike Karl, decided to see whether mice could regenerate their retinas. We found that mice were just like humans in that when the retina was damaged, there was no retinal regeneration, said Dr. Reh. The team then went back to their studies of fish and chicks for clues as to how they could stimulate retinal regeneration in mice. In these animals, glial cells are activated just after the retina is damaged. Unlike neurons, glial cells can go through cell division, so the activated glial cells divide and some of them become new neurons. Dr. Reh and his fellow researchers thought that if they could stimulate glial cells to divide in mice with retinal damage using growth factors, the mice could produce new neurons. Growth factors are proteins normally found in the body that stimulate tissue growth. Some of them are present only in embryonic stages of development and not in adults.
To determine which growth factors to use, Dr. Reh initially looked at previous experiments during embryonic development to find growth factors that may be relevant to neuronal growth during regeneration. Previous research had found that the receptor for epidermal growth factor (EGF) gets turned on when the retina is damaged. Since EGF was associated with retinal development in mammals, Dr. Reh began by testing EGF in mice with retinal damage.
Glial Cells, Again
In the first set of experiments with growth factors in mice, Dr. Rehs team gave mice with retinal damage an injection of EGF. They found that in mice given EGF after retinal damage, glial cells were stimulated to re-enter the mitotic cycle of cell division and divide, growing new cells. This was an exciting finding, but Dr. Reh wanted to know about the effects of other growth factors on glial cells.
Interestingly, fibroblast growth factor (FGF) in combination with insulin, was found by Dr. Rehs laboratory (Fischer et al. 2002) to stimulate Müller glia to dedifferentiate, proliferate, and generate new neurons in posthatch chicken retina, and FGF is known to be important in retinal regeneration in frogs. It seems that FGF and IGF are important in restoring the retina in lots of different vertebrates, remarked Dr. Reh. Using the same experimental set up as before, they tested the ability of FGF and insulin to stimulate glial division in adult mice with retinal damage. They found that the combination of FGF and insulin together were particularly good for the production of neurons in these mice (Karl et al. 2008). In these experiments, Dr. Reh was able to stimulate mice to regenerate some amacrine cells in the retina. This is the first time retinal regeneration has been shown to occur in mice. Dr. Reh was able to stimulate the production of a significant amount of amacrine cells using this treatment. Still, in order for this regeneration to actually repair retinal function that number would need to be increased. Even if this number was increased, it would also be necessary to stimulate the production of other types of neurons as well. Amacrine cells are important in motion detection and serve many other functions such as vision clarity and night vision. Until today they are not, however, associated with any retinal degenerative disease, such as macular degeneration or retinitis pigmentosa, but might be lost secondarily in glaucoma disease.
Genes
The focus of Dr. Rehs current work is to find growth factors to stimulate the production of all types of neurons in the retina. By looking at the regeneration processes in fish and amphibians and embryonic development in mice, Dr. Reh is identifying genes important to these processes and growth factors that may stimulate retinal regeneration. Two genes Dr. Reh has identified are the MASH-1 gene and the PAX6 gene. Both of these genes are needed for retinal regeneration in fish. MASH-1 is released very early in embryonic development and is important for making most retinal cell types. PAX6 is important for all cells except for amacrine cells and is released after MASH-1 but still early in the development process. Through his research, Dr. Reh hopes to unlock the mechanism of retinal regeneration in mammals including, someday, humans.
Dr. Thomas Reh is a Professor of Biological Structure and the Director of Neurobiology and Behavior at the University of Washington School of Medicine in Seattle, Washington. His research focuses on the biological processes of retinal regeneration. Dr. Rehs lab is divided into two groups: one studying retinal development and one studying retinal regeneration. I have always been interested in science, said Dr. Reh, I just never knew exactly what field. In college Dr. Reh heard about retinal regeneration in amphibians and decided that he wanted to study retinal regeneration not only in amphibians but other animal models as well. In his free time, Dr. Reh enjoys skiing, sailing, and mountain climbing.
For More Information:
- Fischer AJ, McGuire CR, Dierks BD, Reh TA. Insulin and fibroblast growth factor 2 activate a neurogenic program in Müller glia of the chicken retina. J Neurosci. 2002 Nov 1;22(21):9387-98.
- Karl MO, Hayes S, Nelson BR, Tan K, Buckingham B, Reh TA. Stimulation of neural regeneration in the mouse retina. Proceedings of the National Academy of Sciences USA. 2008 Dec 9;105(49):19508-13. Epub 2008 Nov 25.
- Lamba D, Karl M, Reh T. Neural regeneration and cell replacement: a view from the eye. Cell Stem Cell. 2008 Jun 5;2(6):538-49. Review.
To Learn More:
Written by Rebecca Kranz with Andrea
Gwosdow, Ph.D. Gwosdow
Associates
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|

Dr. Reh's Lab Team


Watch The Video


Watch The Video
Early multimedia education from nearly 7 decades ago in these two classic 1941 videos about how the human eye functions. An interesting project for you would be to compare the state of knowledge in 1941 with what we know today, and see if any of their information has been superseded by more up-to-date research.
Video courtesy of Youtube.com
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

