Close your eyes for a moment. Imagine going through the entire day with your eyes closed, blinded despite the underlying ability to see. Now imagine living this way, your eyes closed for the unforeseeable future. This is the experience of over 20 million people worldwide who live with a condition known as
cataracts,
an accumulation of misfolded protein in the lens of the eye that obstructs vision. As the cataract grows it can cause total blindness. Cataracts are the leading cause of blindness in the world. Ironically, despite their blindness, most people with cataracts retain the underlying ability to see.
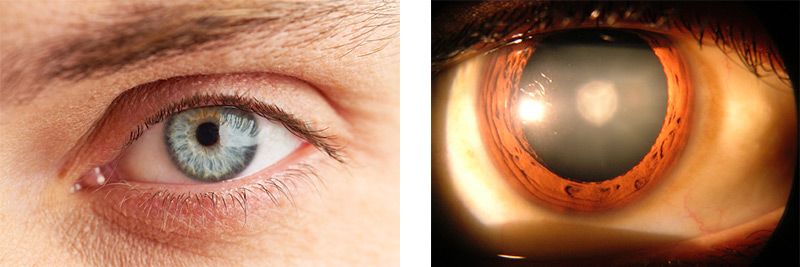
Figure 1. Normal human eye (left) and cataract in human eye (right)
Blindness caused by cataracts is completely treatable. In the United States, more than half of seniors over the age of 70 years have had their vision restored through a minor surgical procedure that removes the cataract. In fact, cataract surgery is the most common surgical procedure in the world, and also one of the safest. Tragically, millions of people around the world do not have access to this life-changing procedure.
The good news is that solutions are on the way. This month we highlight the work of Dr. Jason Gestwicki, Associate Professor of Pharmaceutical Chemistry at the University of California San Francisco, and his former graduate student, Dr. Leah Makley. Dr. Gestwicki and Dr. Makley are developing a non-invasive treatment for cataracts, and the company they founded together, ViewPoint Therapeutics, seeks to further develop the treatment and ultimately deliver it to patients worldwide. Dr. Makley now serves as the Chief Scientific Officer of Viewpoint Therapeutics.
Get ready to open your eyes.

Figure 2. Schematic of the eye
Blinded by Cataracts
Let's begin by exploring the healthy eye. There are three parts of the outer eye that you see daily, whenever you "look someone in the eye." The
cornea
is the clear surface of the eye that allows light to pass through and helps to focus it. The cornea also protects the eye from damage. The black dot in the middle of the eye, called the
pupil,
is an opening that determines the amount of light entering the eye. The colored ring around the pupil, known as the
iris,
regulates the size of the pupil. Located out of sight behind the pupil is the
lens,
a clear tissue that further focuses incoming light before it reaches the back of the eye. The focused light bounces off of the
retina,
a layer of light-sensitive cells in the back of the eye that convert light into electrical signals, which are passed on to the brain via the
optic nerve.
As you can see, healthy vision and healthy functioning of the eye is a complex process. Problems with any one of the steps in this process can lead to limited vision or blindness. For example, the eye disease called
glaucoma
causes vision loss through damage to the optic nerve. Retinal diseases such as
macular degeneration,
on the other hand, affect the ability of the retina to function properly.
Unlike glaucoma or macular degeneration, cataracts do not permanently affect the process of vision. Instead, cataracts prevent vision from taking place by blocking incoming light from reaching the back of the eye. This occurs in the lens, the clear tissue behind the pupil that is composed of water and crystallin proteins dissolved in the water. A cataract forms when the dissolved protein starts to clump together, forming a milky-white aggregate. While this clump of proteins remains small, vision may only be minimally affected. As it grows, however, vision can become blurry, and eventually the cataract will obstruct vision altogether. In this way, cataracts block vision by preventing light from entering the eye. If the cataracts were removed, and light were to regain entry, vision would be restored.
Why cataracts begin to form is still unclear. Babies can sometimes be born with cataracts, and cataracts can form after an acute eye injury, infection, or treatment with certain types of medications. Certain diseases like
diabetes,
personal habits like smoking, and exposure to direct sunlight may accelerate the development of cataracts. Nevertheless, in most cases it appears that cataract formation is a natural part of the aging process.
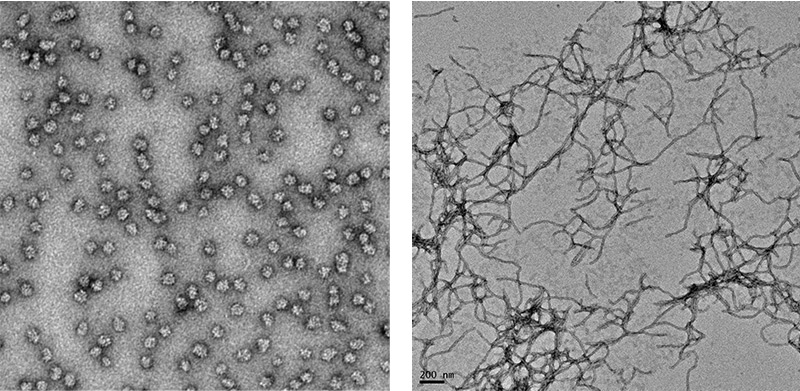
Figure 3. Microscope image of normal protein (left) and misfolded proteins (right)
Cataracts fall into a large class of diseases caused by the misfolding of proteins. Your body makes thousands of proteins every second according to the genetic information contained in your DNA. Each protein must fold into a specific three-dimensional shape in order to function properly. You may be familiar with other diseases caused by misfolded proteins such as
Parkinson's disease
and
Alzheimer's disease.
In the case of cataracts, healthy crystallin proteins in the lens start to change shape so that they are no longer soluble in water. The insoluble form of the protein accumulates in the lens, giving the eye a milky-white color and causing vision to cloud over. As more protein becomes insoluble, the cataract obstructs more and more light until vision is eventually blocked altogether.
One way to think of cataract formation is to imagine making an omelet with egg whites. When you first crack the egg into the frying pan, the egg whites are clear, almost transparent. As the egg whites cook, the heated proteins change shape and stick together, creating the white color that gives egg-whites their name. Cataract formation does not occur through heating, but the change in shape and texture that occurs in the making of an omelet is similar to what happens in the lens.
Re-folding Proteins
Dr. Gestwicki studies the processes involved in protein misfolding in the body. He wanted to know how and why lens proteins change from the soluble healthy state that enables vision to the insoluble unhealthy state of cataracts. In order to gather clues about the process, Dr. Gestwicki and a graduate student working in his lab, Leah Makley, designed a series of experiments to try to reverse the protein aggregation process.
"We were trying to unboil the egg," Dr. Gestwicki explained.
In their experiments, Drs. Gestwicki and Makley relied on the structures of soluble and insoluble proteins. They realized that the insoluble protein was actually the more stable structure. This meant that more energy—in the form of heat—would be required to unfold the insoluble protein, resulting in a higher melting point. Indeed, the researchers identified the melting point of the soluble protein to be about 65 degrees Celsius, whereas the insoluble protein had a melting point that was significantly higher, about 70 degrees Celsius. This was the quantifiable characteristic that Drs. Gestwicki and Makley used to find molecules that would convert the insoluble protein back into its soluble form.
Drs. Gestwicki and Makley began with approximately 2,500 molecules to test for stabilization effects on the insoluble protein, indicated by a reduction in the melting point. In order to conduct the same experiment on so many molecules, the researchers used microwell plates, small plates about the size of a 4 x 6 inch index card that contained 384 small wells. Each well was filled with the same amount of insoluble protein, and then a small volume of a solution containing a test molecule was added. The microwell plate was placed in an instrument that slowly heated the contents of the plate, allowing the researchers to identify the melting point of any individual well. In this way, the researchers conducted 384 mini-experiments simultaneously, a process known as
high throughput screening.

Figure 4. 384-well plate
These experiments continued until a drop in melting point occurred, indicating that the molecule might improve the solubility of the protein. Of the 2,500 molecules the researchers began with, 60-80 were identified as having some stabilization effect. After validation experiments, however, only 30 molecules remained.
The next step was to examine the structure of each potential molecule. The researchers found that twelve of the thirty molecules had basically the same structure, that of an oxysterol molecule, in the same class of lipid molecules as
cholesterol.
"The biology was screaming at us. There was something important going on with that type of molecule."
Next, Drs. Gestwicki and Makley spent about a year improving upon the structure of the oxysterols they had identified in the high throughput screening experiments. Using their knowledge of drug chemistry, the researchers set out to create a molecule that would be as safe and effective as possible. The goal was to deliver this compound directly to the eye with an eye dropper so that it would stabilize the insoluble protein that was causing vision problems, ultimately restoring vision. Finally, the researchers identified the most soluble and active compound for use in further experiments: Compound 29.
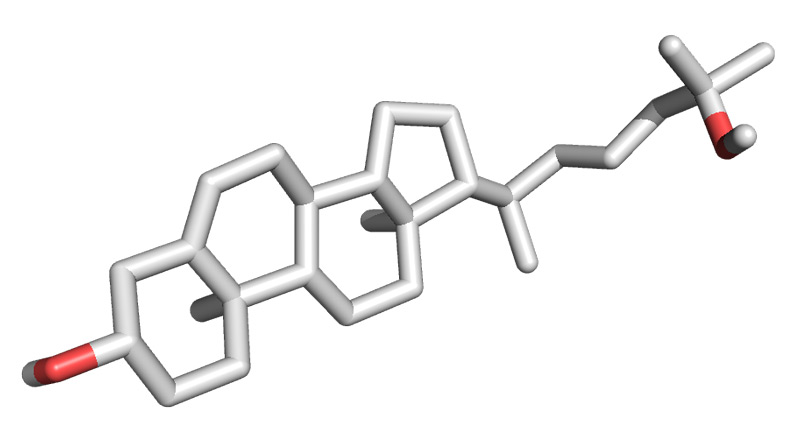
Figure 5. 3D oxysterol
Having identified a potential therapeutic compound in cellular (in vivo) experiments, the next step was to conduct trials using living organisms (in vivo). To do this, the researchers used a
mouse model
of cataract formation. The animals had an
inherited
form of cataracts that developed with equal severity in both eyes after just a few weeks of life. In one cloudy eye the researchers placed a control solution commonly found in eye drops. In the other, the researchers placed the control solution containing Compound 29. Then they checked the mice each day. "The results were astounding," recalled Dr. Gestwicki. "After two weeks we could tell which eye had been treated by Compound 29 and which hadn't from across the room!" That is because the untreated eye remained cloudy while the treated eye became clear. "After this experiment, we knew we were on to something," Dr. Makley added.
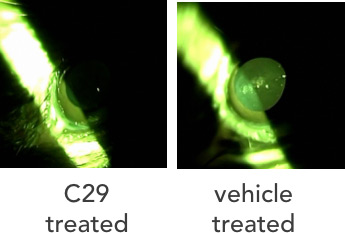
Figure 6. Mouse eye treated with Compound 29 (left) and untreated (right)
In addition to their physical observations, the researchers measured the amount of soluble and insoluble protein in each eye. These experiments confirmed that Compound 29 was behaving just as the researchers had hoped—stabilizing the misfolded protein so that it became soluble again. "This was a happy day in science," commented Dr. Gestwicki. "Compound 29 showed real promise."
From the Laboratory to Market
In order to make Compound 29 available for patient use, Drs. Gestwicki and Makley founded a company called ViewPoint Therapeutics in San Francisco, CA. As Chief Scientific Officer, Dr. Makley now oversees all of the experiments necessary to bring this drug to market. This includes safety and efficacy studies to determine the optimal amount of drug to deliver with the fewest side effects. This process can take up to a decade, although Dr. Makley hopes it won't take quite that long. "This is such an important problem," remarked Dr. Makley, "and we have a better way to solve it. We want to make a safe and effective treatment available to patients around the world as quickly as possible."
Dr. Gestwicki remains involved with ViewPoint Therapeutics, and his laboratory research has moved on to continue to study other diseases caused by protein misfolding such as Parkinson's disease and Alzheimer's disease. "In many ways cataracts are a much simpler, more straightforward problem," Dr. Gestwicki commented. "But our success with cataracts has really encouraged us to tackle these complex brain diseases."
Dr. Jason Gestwicki is Associate Professor of Pharmaceutical Chemistry at the University of California San Francisco and co-founder of ViewPoint Therapeutics. His research focus includes the chemistry of protein misfolding diseases such as cataracts, Parkinson's disease, and Alzheimer's disease. When not in the laboratory, Dr. Gestwicki enjoys listening to, researching and writing about new music, and spending time with his family.
Dr. Leah Makley is Chief Scientific Officer for ViewPoint Therapeutics in San Francisco, CA. She received her PhD in Medicinal Chemistry from the University of Michigan under the supervision of Dr. Gestwicki and wrote her doctoral dissertation in part about this research treatment for cataracts. When not at the office or laboratory, Dr. Makley enjoys running and playing the piano.
ViewPoint Therapeutics is a small biotechnology company founded by Drs. Gestwicki and Makley in 2014. The mission of the company is to deliver drug therapies that address diseases caused by protein misfolding. The company is currently focused on using Compound 29 as a starting point to develop a non-surgical treatment for cataracts and developing new and improved molecules that work in the same way.
For More Information:
- Makley, L. et al. 2015. “Pharmacological chaperone for α-crystallin partially restores transparency in cataract models.” Science, 350(6261): 674-7.
- Quinlan, R. 2015. “A new dawn for cataracts.” Science, 350(6261): 636-7.
To Learn More:
- Viewpoint Therapeutics.
http://www.viewpointtherapeutics.com
- Gestwicki Laboratory.
http://gestwickilab.ucsf.edu/
- National Eye Institute.
https://nei.nih.gov/health/cataract/cataract_facts
- World Health Organization.
http://www.who.int/topics/cataract/en/
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com

