| |
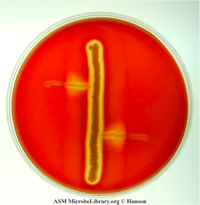
Staphylococcus aureus streaked on a
blood agar plate
Did you know that the human body contains approximately ten times more
bacterial cells than human cells? These bacteria comprise more than 500
different species and can weigh several pounds. Fortunately, most of these
bacteria are beneficial to your body, or at least not harmful. For example,
Lactobacillus and
Bifidobacteria bacteria, commonly found in food products such as yogurt as well as in the human gut, aid in digestion.
Escherichia coli, which is also found in the gut and aids in digestion, is another example of a beneficial bacterial species in normal populations. But E. coli can also cause infection if found somewhere other than the gut or if the population increases to an abnormal size. Of course, there are also many bacteria that are foreign to the human body and can cause infection when they get inside. The role of the
immune system is to prevent and fight such infections.
In order to cause infection, pathogenic bacteria must be able to obtain all the nutrients they need from the human body, including metals such as zinc, iron, and manganese. As a result, many vertebrate species (including humans) have evolved ways to store these metals in forms that are not readily accessible to bacteria. This mechanism is known as nutritional immunity.
Locking the Iron Away
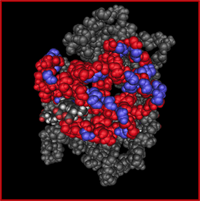
The structure of hemoglobin
Iron is one of the most difficult nutrients for bacteria to get from the human body.
Most of the iron found in the human body is not readily available in a form bacteria
can use. About 80% of iron in the body is found in the
hemoglobin of
red blood cells Hemoglobin is responsible for the crucial task of transporting oxygen throughout the bloodstream. The basic structure of the hemoglobin molecule contains four heme cofactors. Heme is a circular molecule with an iron atom at the center. Each iron atom of heme binds one oxygen atom. Therefore, hemoglobin contains four iron atoms which bind four atoms of oxygen.
In order to obtain iron inside the human body,
then, a bacterium would have to find a way to extract iron from the hemoglobin inside red blood cells.
The bacterium
Staphylococcus aureus has such a mechanism. S. aureus bacteria obtain iron from within the red blood cells by producing toxins that destroy red blood cells. These toxins cause the cell membrane of red blood cells to disintegrate, releasing the cells components, including hemoglobin, into the surrounding fluid, in a process known as
lysis.

The periodic table for iron
The bacteria then capture hemoglobin from the lysed cells and extract the iron from inside the hemoglobin molecule. The process of extracting iron from red blood cells is essential to the survival of S. aureus bacteria. It is also what makes S. aureus so successful at causing infections.
Dangerous Staphylococcus
S. aureus is one of the deadliest bacteria to humans, causing diseases ranging from minor skin infections and boils to
pneumonia,
meningitis,
endocarditis,
toxic shock syndrome, and
sepsis.
S. aureus infections, commonly known as staph infections, are the leading cause of hospital-acquired infections in the United States. Over 40,000 people die annually in the United States as a result of staph infections. In addition to infecting humans, S. aureus also causes udder infection in cattle known as
bovine mastitis,
and similar infections in pigs and sheep. A better understanding of this organism and the process of iron extraction is therefore essential to treating and preventing staph infections.
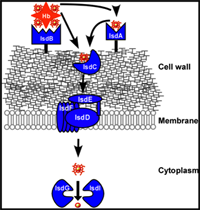
Staphylococcus aureus Isd system
Dr. Eric Skaar of Vanderbilt University Medical Center in Nashville, Tennessee has been studying the S. aureus bacteria for over a decade. Several years ago, Dr. Skaar and his team of researchers described the pathway involved in the extraction of iron by S. aureus. This pathway, known as the iron-regulated surface determinant system, or Isd system, involves around twelve S. aureus genes encoding for many different proteins and enzymes. Of particular interest to Dr. Skaar was the protein known as the IsdB receptor, which binds to the exterior of the hemoglobin molecule as soon as the red blood cells burst. Although the inner amino acids of the hemoglobin molecule are very similar across species, the outer amino acids are more variable. Dr. Skaar hypothesized that differences in the outside portion of the hemoglobin would impact the ability of S. aureus to bind to hemoglobin and extract iron.
Putting a Hypothesis to the Test
To test his hypothesis, Dr. Skaar conducted two sets of experiments using blood samples from a variety of different species including humans, baboons, pigs, cows, horses and sheep. The overall goals of these experiments were to determine whether 1) S. aureus could bind the same amount of hemoglobin in all species, and 2) if bacteria could bind more easily to some types of hemoglobin than others.
In the first set of experiments, Dr. Skaar and his student Gleb Pishchany measured the ability of S. aureus to bind to the hemoglobin of different animal species. To begin, they first extracted purified hemoglobin from the blood samples through a process called
centrifugation. Centrifugation takes advantage of gravity to separate materials of different masses, and is very useful for separating different molecules found in blood. Vials of blood are placed on a machine called a
centrifuge that spins very quickly, causing red blood cells to settle to the bottom of the vial. Red blood cells are then lysed and hemoglobin can be extracted from the centrifuged sample.
Next, Dr. Skaar and Gleb Pishchany placed the hemoglobin from different animal species into different vials with S. aureus cells. The S. aureus cells that were able to recognize the hemoglobin would tightly bind to it, resulting in three types of molecules in the mixture: hemoglobin, S. aureus cells, and S. aureus cells bound to hemoglobin. Again the vials were placed in the centrifuge to separate all the S. aureus cells from the unbound hemoglobin molecules and the S. aureus cells were extracted from the mixture. Dr. Skaar then measured the amount of hemoglobin that bound to S. aureus for each species. These results showed distinct differences in the ability of S. aureus to use the hemoglobin of different species. Dr. Skaar found that S. aureus was best able to bind human hemoglobin. He also noticed a trend that the S. aureus bacteria could bind more effectively hemoglobin from species more closely related to humans evolutionarily.
In the second set of experiments, Dr. Skaar and Gleb Pishchany measured the ability of S. aureus cells to grow using iron obtained from the hemoglobin of different animal species. They first cultured S. aureus cells on a medium that lacked iron and therefore inhibited cell growth. Next he added hemoglobin from different animal species to the medium as the only iron source and measured the growth area of the bacteria. A larger growth area would indicate better, more efficient use of the iron source. Similar to the first set of experiments, this growth experiment demonstrated that S. aureus bacteria use human hemoglobin most effectively as an iron source when compared to the other animal species tested. Dr. Skaar observed the bacteria were able to grow better using hemoglobin from species more closely related to humans evolutionarily.
Animals Differ, People Differ
Variations in the ability of S. aureus to bind hemoglobin among species suggest that there may also be variations within the human population that would make some people more susceptible to staph infection than others. We know that about one-third of the worlds population has S. aureus bacteria living in their noses, but not all of these people develop staph infections.
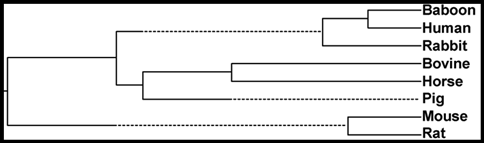
A dendrogram showing the relatedness of hemoglobins across animals
Dr. Skaar believes there may be genetic differences within the gene that encodes for human hemoglobin that predispose some people to staph infection and not others. He is just beginning a genetic study to identify which genetic sequences of the hemoglobin gene may make some patients more susceptible to staph infections than others. Dr. Skaar and his team are examining the genetic information of hospital patients who develop staph infections to identify particular sequences of the hemoglobin gene they may have in common that are distinct from the sequences of the rest of the population. By comparing the genetic sequences of patients who develop staph infection to those who do not, the researchers hope to determine whether there are certain hemoglobin sequences that would predispose patients to staph infections. If this is the case, it would then be possible for doctors to identify patients that are at risk for staph infections before they enter a hospital setting. These patients could potentially be treated prior to admission into the hospital, and special precautions could be made to prevent infection.
Dr. Eric Skaar is Associate Professor of
Microbiology and
Immunology at the Vanderbilt University Medical Center in Nashville, Tennessee. Dr. Skaar studies the host-pathogen interaction with specific focus on the Staphylococcus aureus pathogen with the ultimate goal of developing new treatments for infectious diseases. When not in the laboratory, Dr. Skaar enjoys running, golfing, and spending time with his family.
For More Information:
- Anzaldi, L. and E. Skaar. 2010. "Overcoming the Heme Paradox: Heme Toxicity and Tolerance in Bacterial Pathogens." Infection and Immunity, 78(12): 4977-4989.
- Kehl-Fie, Thomas. 2009. "Nutritional immunity beyond iron: a role for manganese and zinc." Current Options in Chemical Biology, 14: 1-7.
- Reniere, M., V. Torres, E. Skaar. 2007. "Intracellular metalloporphyrin metabolism in Staphylococcus aureus." Biometals, 20: 333-45.
To Learn More:
Rebecca Kranz with Andrea Gwosdow, Ph.D. Gwosdow Associates
A note about Wikipedia from Wikipedia: "Wikipedia cannot guarantee the validity of the information found here. The content of any given article may recently have been changed, vandalized or altered by someone whose opinion does not correspond with the state of knowledge in the relevant fields. Note that most other encyclopedias and reference works also have similar disclaimers." http://en.wikipedia.org/wiki/Wikipedia:General_disclaimer.
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |
|
|

Dr. Erick Skaar and graduate student Gleb Pishchany
Photo: Susan Urmy/Vanderbilt University Medical Center
|

The Skaar Lab team at Vanderbilt University
|
Sign Up for our Monthly Announcement!
...or  subscribe to all of our stories! subscribe to all of our stories!

What A Year! is a project of the Massachusetts
Society for Medical Research.
|
|

