Highlights
These researchers created an antimicrobial bone-healing hydrogel to simultaneously treat bone infection and promote healing. They combined the antimicrobial enzyme lysostaphin with bone morphogenetic protein-2 (BMP-2). Lysostaphin treats the infection and BMP-2 stimulates bone growth. The researchers showed that lysostaphin significantly reduced, and in some cases completely eliminated, the bone infection when delivered through the hydrogel. Lysostaphin did not affect the immune system of mice and did not damage the liver.
Across the globe, the widespread use of
antibiotics
has led to an increasingly pervasive threat: antibiotic-resistant bacteria. The first antibiotic,
penicillin,
was developed in 1928 – less than a century ago. Since that time, antibiotics have been used to treat all types of infections in humans and animals – so much so that many of these bacteria have developed resistance to common antibiotics.
The Centers for Disease Control and Prevention
estimate that about two million people each year contract an antibiotic-resistant infection, causing death in at least 35,000 of these cases.
A global response is necessary to combat antibiotic resistance including reducing the use of antibiotics and developing more effective treatments for infections and antibiotic resistance. The best way to reduce antibiotic use is to prevent infections in the first place.
Dr. Andrés García has witnessed firsthand the consequences of antibiotic resistance. Dr. García is the Executive Director of the Parker H. Petit Institute for Bioengineering and Bioscience at Georgia Institute of Technology. His research has focused on engineering biomaterials for the therapeutic delivery of proteins and cells. This focus expanded when his wife was hospitalized after her appendix burst and she needed emergency surgery. Thankfully she recovered from the surgery, only to develop an infection at the surgical site. She was treated with regular antibiotics that cleared the initial infection, but then she developed a second antibiotic-resistant infection. She was in and out of the emergency room for four months before her doctors found a treatment that worked.
This whole experience stimulated Dr. García’s thinking about how he could use his expertise in engineering and biomaterials to develop a drug delivery system to prevent and treat infection. His laboratory had collaborated in the past with
orthopedists
and
microbiologists
to create drug delivery systems for bone repair. Now, Dr. García wanted to develop a system that would fight and prevent infection while also promoting bone healing. To do this, he collaborated with Dr. Lars Westblade at Emory University (now at Weill Cornell Medicine) and Dr. Rod Donlan from the Centers for Disease Control and Prevention to gain expertise in microbiology. In the García lab, graduate students Chris Johnson and Jeremy Caplin and post-doctoral fellow Dr. Pranav Kalelkar were involved in this research.
One of the main risks of orthopedic surgery is infection that develops in the bone. Even though surgeons do their best to work in a sterile environment with sterile materials, bone is supposed to remain protected inside the body. Exposure to the outside world, in addition to any hardware like screws that need to be implanted, opens the possibility for
infection,
which occurs in approximately 30% of people with open fractures.
Bone infections can be particularly difficult to treat. They often require additional surgeries, and they may cause any implanted hardware to fail, resulting in further procedures. In addition, bone infections slow down the healing process. Dr. García and his colleagues wanted to address these issues with bone infections by creating a material that would simultaneously treat the bone infection and promote healing.
The delivery system that Dr. García and his team used was one they were already familiar with in their years of research experience:
hydrogels.
Hydrogels are water swollen crosslinked
polymers
, meaning that they are made up of repeating smaller units. Each unit has four arms that are chemically prompted to link with each other, creating a mesh-like material. The mesh then swells with water, resulting in a hydrogel that is about 5% polymer and 95% water. One hydrogel you may have heard of is jello: a material that is mostly water contained by a mesh polymer, one that happens to taste quite good.

Figure 1. Hydrogel in implantation tub
[Credit: GT Institute Communications]
The most common type of bone infection is caused by the bacteria Staphylococcus aureus. An effective treatment for this infection is the
enzyme
lysostaphin, produced by a similar type of bacteria. Although it is effective, lysostaphin is difficult to stabilize and therefore is not currently available to treat infections. Dr. García and his team chose lysostaphin because the hydrogel material stabilizes the compound and could create a mechanism to deliver this treatment to the site of infection.
To promote bone healing, Dr. García and his team used
bone morphogenetic protein
(BMP) that had already been approved by the Food and Drug Administration for use in humans. In particular, bone morphogenetic protein 2 or BMP-2 was known to improve bone formation.
To create this antimicrobial bone-healing hydrogel, the researchers used the same process that they had already developed to create hydrogels. The difference was that when they induced the chemical reaction to link the smaller units into a polymer, they also added lysostaphin and BMP-2. This way the two compounds became part of the hydrogel.
Once the hydrogel had been developed, Dr. García and his team began experiments. First, they tested the release of the antimicrobial and protein from the hydrogel. To do this, they created a hydrogel with fluorescent lysostaphin and BMP-2 so they could watch and quantify the molecules being released. They also tested the release of lysostaphin with an additional molecule called collagenase that degrades the hydrogel, increasing the rate of release of the lysostaphin. The goal of these initial experiments was to optimize the concentration and release rates of lysostaphin and BMP-2.
With these initial experiments, the researchers determined that the hydrogel they developed would release lysostaphin and BMP-2 as intended. Next, Dr. García and his team used mouse models to determine the effectiveness of the material they developed. They applied the hydrogel to mice with bone injuries who had developed S. aureus infection. The researchers were able to show that lysostaphin significantly reduced, and in some cases completely eliminated, the infection when delivered through the hydrogel. Similarly, Dr. García and his team showed that BMP-2 promoted healing and improved bone strength in injured bones.
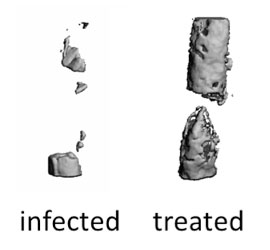
Figure 2. Bone infected and bone treated with hydrogel containing lysostaphin and BMP-2.
[Credit: Dr. Garcia]
The results in mouse models were very promising. Before they could call the hydrogel a success in mice, though, the researchers needed to test the reaction of the mouse’s immune system. Since lysostaphin does not naturally occur in the mouse, it could trigger an immune response, even as it helped clear the infection. In addition, bacterial debris after lysostaphin killed the bacteria could cause inflammation. To test the immune response, the researchers analyzed markers of inflammation in the bloodstream including liver enzymes. The results showed that lysostaphin did not provoke an immune response and was not damaging to the liver.
“These results are very promising,” remarked Dr. García. “We hope these hydrogels are able to help many people with bone injuries.” There are still many more experiments to conduct before the hydrogel can be tested in humans. The next step is to test the material in a larger animal model. Dr. García and his team are currently conducting experiments in sheep that are similar to those described here. If those experiments are successful, they can work towards getting the material approved for veterinary use.
Dr. Andrés García is Executive Director of the Parker H. Petit Institute for Bioengineering and Bioscience at Georgia Institute of Technology. His research focuses on tissue engineering and the creation of biomaterials for drug delivery systems. When not in the laboratory, Dr. García enjoys sports, reading, spending time outdoors, and spending time with his family.
For More Information:
- Johnson, C. et al. 2019. “Lysostaphin and BMP-2 co-delivery reduces S. aureus infection and regenerates critical-sized segmental bone defects.” Science Advances, 5: eaaw1228. https://advances.sciencemag.org/content/5/5/eaaw1228/tab-pdf
- Caplin J. and A. García. 2019. “Implantable antimicrobial biomaterials for local drug delivery in bone infection models.” Acta Biomaterialia, 93: 2-11. https://www.sciencedirect.com/journal/acta-biomaterialia/vol/93
- Johnson, C. et al. 2018. “Hydrogel delivery of lysostaphin eliminates orthopedic implant infection by Staphylococcus aureus and supports fracture healing.” Proceedings of the National Academy of Sciences, 115 (22): E4960-69. https://www.pnas.org/content/115/22/E4960.long
To Learn More:
- García Lab. http://garcialab.gatech.edu/
- World Health Organization. https://www.who.int/news-room/fact-sheets/detail/antibiotic-resistance
- United States Centers for Disease Control and Prevention. https://www.cdc.gov/drugresistance/about.html
- Ventola, C. 2015. “The Antibiotic Resistance Crisis.” https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4378521/
Written by Rebecca Kranz with Andrea Gwosdow, PhD at www.gwosdow.com
HOME | ABOUT | ARCHIVES | TEACHERS | LINKS | CONTACT
All content on this site is © Massachusetts
Society for Medical Research or others. Please read our copyright
statement — it is important. |

